��Ŀ����
��ͬ�¶��£��������ȵ����������ܱ������з������淴Ӧ��
2NH3(g)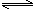 N2(g)+3H2(g) ��H��+92.4kJ/mol
N2(g)+3H2(g) ��H��+92.4kJ/mol
ʵ������ʼ��ƽ��ʱ���й��������±���
| ������� | ��ʼʱ���������ʵ���/mol | ƽ��ʱ��Ӧ�е������仯 | ||
| NH3 | N2 | H2 | ||
| �� | 2 | 0 | 0 | ��������akJ |
| �� | 0 | 1 | 3 | �ų�����bkJ |
| �� | 4 | 0 | 0 | ��������ckJ |
����������ȷ����
A��������ϵ��a��b
B����Ӧ��ƽ�ⳣ�����ۣ��٣���
C����ƽ��ʱH2������������٣���
D�����е��ܶȲ��ٸı�ʱ˵����Ӧ�Ѵﵽƽ��״̬
��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ


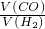 �T______��
�T______��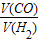 �T______��
�T______��