��Ŀ����
ʵ��С���ͬѧ��ϡ����ⶨijͭп�Ͻ���п����������������������з��������㣮
��1������ͬѧȡ10g�Ͻ������Թ��У������м���10gϡ���ᣬ��ַ�Ӧ���ռ���0.06gH2���ټ���10gϡ�������ռ���0.06gH2���������μ���ϡ����ʱ���ٲ������ݣ���10g�Ͻ���ϡ���ᷴӦ������H2______g��
��2��ʵ��С���ͬѧ�ܿ�������ͭп�Ͻ���п����������Ϊ39%���������ϡ���������ʵ�����������
�⣺��1��10g�Ͻ���ϡ���ᷴӦ����������������Ϊ��0.06g+0.06g=0.12g��
���0.12��
��2����20gϡ���������������ΪX��
Zn+H2SO4�TZnSO4+H2����
98 2
X 0.12g
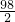 =
= ��
��
X=5.88g��
ϡ���������ʵ���������Ϊ��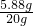 ��100%=29.4%��
��100%=29.4%��
��ϡ���������ʵ�����������29.4%��
��������1���������ɵ������������ͼ�Ϊ����������������
��2�����������������������Լ���μӷ�Ӧ���������������һ�����Լ���ϡ���������������
������������Ҫ����ѧ�����ü��跨�ͻ�ѧ����ʽ���м�����ƶϵ�������
���0.12��
��2����20gϡ���������������ΪX��
Zn+H2SO4�TZnSO4+H2����
98 2
X 0.12g
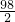 =
= ��
��X=5.88g��
ϡ���������ʵ���������Ϊ��
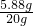 ��100%=29.4%��
��100%=29.4%����ϡ���������ʵ�����������29.4%��
��������1���������ɵ������������ͼ�Ϊ����������������
��2�����������������������Լ���μӷ�Ӧ���������������һ�����Լ���ϡ���������������
������������Ҫ����ѧ�����ü��跨�ͻ�ѧ����ʽ���м�����ƶϵ�������

��ϰ��ϵ�д�
 ѧҵ����һ��һ��ϵ�д�
ѧҵ����һ��һ��ϵ�д�
�����Ŀ
 ijУ��ѧ����С���ͬѧ��ҪһЩ��������ѧʵ�飮����һƿδ������Ũ���ᣬ��ǩ��ͼ�����Ķ���ǩ�ϵ�˵�����ش��������⣮
ijУ��ѧ����С���ͬѧ��ҪһЩ��������ѧʵ�飮����һƿδ������Ũ���ᣬ��ǩ��ͼ�����Ķ���ǩ�ϵ�˵�����ش��������⣮
��1����װ��Ũ������Լ�ƿһ��ʱ�������Һ�����������ӣ���������������______�������С��������ΪŨ�������______�ԣ�
��2��ϡ��Ũ����ʱ����ȷ�IJ�����______����ѡ�
A����Ũ������������ʢˮ���ձ��У����ò��������Ͻ���
B����ˮ����ʢŨ������ձ��У����ò��������Ͻ���
��3���������Է���������______����������Ԫ������Ԫ�ص�������Ϊ______
��4��ȡ����Ũ����50mL�����Ƴ�Ũ��Ϊ19.6%��ϡ���ᣬ��Ҫ��ˮ���ٿˣ�
��5������С���ͬѧ��19.6%��ϡ������ͭп�Ͻ�Ӧ���ⶨͭп�Ͻ���п������������ʵ�����������ʾ��
| ʵ����� | 1 | 2 | 3 |
| �Ͻ�������g�� | 10 | 10 | 10 |
| ϡ����������g�� | 25 | 50 | 75 |
| ����������������g�� | 0.1 | 0.2 | 0.2 |
 ijУ��ѧ����С���ͬѧ��ҪһЩ��������ѧʵ�飮����һƿδ������Ũ���ᣬ��ǩ��ͼ�����Ķ���ǩ�ϵ�˵�����ش��������⣮
ijУ��ѧ����С���ͬѧ��ҪһЩ��������ѧʵ�飮����һƿδ������Ũ���ᣬ��ǩ��ͼ�����Ķ���ǩ�ϵ�˵�����ش��������⣮
 A��ij���������᳧���豸��ª�������¾ɣ��ó�ÿ���ŷŴ�����SO2�ķ����ͺ�H2SO4�����Է�ˮ�����ص����������;������ú̿��ȼ�ϣ�ֻҪ����������꣬�Ը�����ɼ����ƻ���
A��ij���������᳧���豸��ª�������¾ɣ��ó�ÿ���ŷŴ�����SO2�ķ����ͺ�H2SO4�����Է�ˮ�����ص����������;������ú̿��ȼ�ϣ�ֻҪ����������꣬�Ը�����ɼ����ƻ���