��Ŀ����
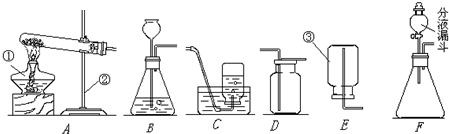
��ͼ��ʵ������ȡ����ͼ����й����ʵ�һЩװ�ã���ش��й�����

��1 ����д��ͼ�����������Ƣ� ���� ���� ��
��2 ��ʵ���ҳ���װ��A ��ȡ��������Ӧ�Ļ�ѧ����ʽΪ ���Թܿڷ�һ������������ ���ռ������ʱ���� ���������� ����ʱ��˵������ƿ�����ռ������塣
��3 ����B ��C װ����ȡ������̼�Ļ�ѧ����ʽΪ �������������������Ĵ�С����B ��C ��ȵ������Т� ���� ��
��2 ��ʵ���ҳ���װ��A ��ȡ��������Ӧ�Ļ�ѧ����ʽΪ ���Թܿڷ�һ������������ ���ռ������ʱ���� ���������� ����ʱ��˵������ƿ�����ռ������塣
��3 ����B ��C װ����ȡ������̼�Ļ�ѧ����ʽΪ �������������������Ĵ�С����B ��C ��ȵ������Т� ���� ��
(1)�� �ƾ��� �� ����̨ �� ����©��
(2) 2KMnO4 K2MnO4+MnO2+O2���� ��ֹҩƷ���뵼�� ��ˮ���������������ȵ�ð��ˮ����������ð����ֻ����ƿ��û��ˮ������ȷ��
K2MnO4+MnO2+O2���� ��ֹҩƷ���뵼�� ��ˮ���������������ȵ�ð��ˮ����������ð����ֻ����ƿ��û��ˮ������ȷ��
(3) CaCO3+2HCl=CaCl2+H2O+CO2�� ���ٿ�����ʱ����Һ��ҩƷ�� �ڿ��Է�ֹ��Ӧ��������ĭ���뵼�ܻ������ȡ�϶������ ( ����������Ҳ��)
(2) 2KMnO4
 K2MnO4+MnO2+O2���� ��ֹҩƷ���뵼�� ��ˮ���������������ȵ�ð��ˮ����������ð����ֻ����ƿ��û��ˮ������ȷ��
K2MnO4+MnO2+O2���� ��ֹҩƷ���뵼�� ��ˮ���������������ȵ�ð��ˮ����������ð����ֻ����ƿ��û��ˮ������ȷ��(3) CaCO3+2HCl=CaCl2+H2O+CO2�� ���ٿ�����ʱ����Һ��ҩƷ�� �ڿ��Է�ֹ��Ӧ��������ĭ���뵼�ܻ������ȡ�϶������ ( ����������Ҳ��)

��ϰ��ϵ�д�
 ��ǰ����ϵ�д�
��ǰ����ϵ�д�
�����Ŀ


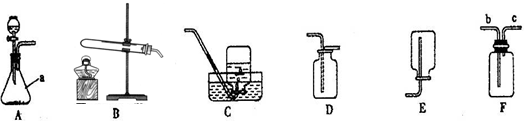
 ��ͼ��ʵ������ȡ����ʱ���õ�װ�ã���ش��������⣮
��ͼ��ʵ������ȡ����ʱ���õ�װ�ã���ش��������⣮
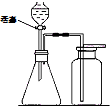 ������ɣ��ȹرջ��������ڼ���ƿ�м�ˮ��û����ĩ�ˣ�Ȼ��
������ɣ��ȹرջ��������ڼ���ƿ�м�ˮ��û����ĩ�ˣ�Ȼ��