��Ŀ����
Ϊ�ⶨij�������ġ��߸�Ƭ�� ��Ʒ��̼��Ƶ���������������������ʵ�飺ȡ11.0g��Ʒ�����ձ��У��Ƶ��ձ�����ʢ��Ʒ��������Ϊ158.0g���ٰ�100gϡ����ƽ���ֳ��ķ����μ�����Ʒ�У�ÿ�ξ���ַ�Ӧ����֪��Ʒ���������ʲ������ᷴӦ����ʵ�����ݼ�¼���£�
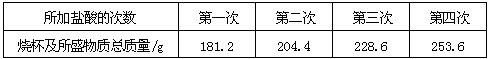
����ݴ˼��㣺
��1����һ�����ɶ�����̼�������� g��
��2�����߸�Ƭ�� ��Ʒ��̼��Ƶ����������Ƕ��٣��������ȷ��0.1%��
��1����һ�����ɶ�����̼�������� g��
��2�����߸�Ƭ�� ��Ʒ��̼��Ƶ����������Ƕ��٣��������ȷ��0.1%��
��1��1.8��
��2���⣺��Ʒ��ȫ��Ӧ����CO2��������158.0g+100g��-253.6g=4.4g
��11.0g��Ʒ��̼�������ΪX����
CaCO3+2HCl===CaCl2+H2O+CO2��
100 44
X 4.4g
100��44=X��4.4g
X=��100��4.4g����44=10g
�߸�Ƭ����Ʒ��̼��Ƶ�����������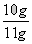 ��100����90.9��
��100����90.9��
�𣺡��߸�Ƭ����Ʒ��̼��Ƶ�����������Ϊ90.9%��
��2���⣺��Ʒ��ȫ��Ӧ����CO2��������158.0g+100g��-253.6g=4.4g
��11.0g��Ʒ��̼�������ΪX����
CaCO3+2HCl===CaCl2+H2O+CO2��
100 44
X 4.4g
100��44=X��4.4g
X=��100��4.4g����44=10g
�߸�Ƭ����Ʒ��̼��Ƶ�����������
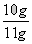 ��100����90.9��
��100����90.9���𣺡��߸�Ƭ����Ʒ��̼��Ƶ�����������Ϊ90.9%��

��ϰ��ϵ�д�
�����Ŀ
Ϊ�ⶨij�������ġ��߸�Ƭ����Ʒ��̼��Ƶ���������������������ʵ�飺ȡ11.0g��Ʒ�����ձ��У��Ƶ��ձ�����ʢ��Ʒ��������Ϊ158.0g���ٰ�100gϡ����ƽ���ֳ��ķ����μ�����Ʒ�У�ÿ�ξ���ַ�Ӧ����֪��Ʒ���������ʲ������ᷴӦ����ʵ�����ݼ�¼���£�
����ݴ˼��㣺��1����һ�����ɶ�����̼�������� g��
��2�����߸�Ƭ����Ʒ��̼��Ƶ����������Ƕ��٣��������ȷ��0.1%��
| ��������Ĵ��� | ��һ�� | �ڶ��� | ������ | ���Ĵ� |
| �ձ�����ʢ����������/g | 181.2 | 204.4 | 228.6 | 253.6 |
��2�����߸�Ƭ����Ʒ��̼��Ƶ����������Ƕ��٣��������ȷ��0.1%��
Ϊ�ⶨij�������ġ��߸�Ƭ����Ʒ��̼��Ƶ���������������������ʵ�飺ȡ11.0g��Ʒ�����ձ��У��Ƶ��ձ�����ʢ��Ʒ��������Ϊ158.0g���ٰ�100gϡ����ƽ���ֳ��ķ����μ�����Ʒ�У�ÿ�ξ���ַ�Ӧ����֪��Ʒ���������ʲ������ᷴӦ����ʵ�����ݼ�¼���£�
| ��������Ĵ��� | ��һ�� | �ڶ��� | ������ | ���Ĵ� |
| �ձ�����ʢ����������/g | 181.2 | 204.4 | 228.6 | 253.6 |
��2�����߸�Ƭ����Ʒ��̼��Ƶ����������Ƕ��٣��������ȷ��0.1%��
Ϊ�ⶨij�������ġ��߸�Ƭ����Ʒ��̼��Ƶ���������������������ʵ�飺ȡ11.0g��Ʒ�����ձ��У��Ƶ��ձ�����ʢ��Ʒ��������Ϊ158.0g���ٰ�100gϡ����ƽ���ֳ��ķ����μ�����Ʒ�У�ÿ�ξ���ַ�Ӧ����֪��Ʒ���������ʲ������ᷴӦ����ʵ�����ݼ�¼���£�
����ݴ˼��㣺��1����һ�����ɶ�����̼��������______g��
��2�����߸�Ƭ����Ʒ��̼��Ƶ����������Ƕ��٣��������ȷ��0.1%��
| ��������Ĵ��� | ��һ�� | �ڶ��� | ������ | ���Ĵ� |
| �ձ�����ʢ����������/g | 181.2 | 204.4 | 228.6 | 253.6 |
��2�����߸�Ƭ����Ʒ��̼��Ƶ����������Ƕ��٣��������ȷ��0.1%��