��Ŀ����
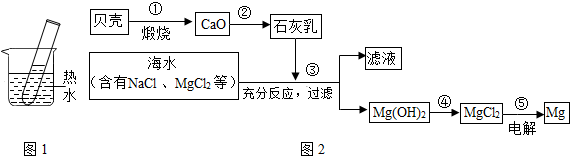
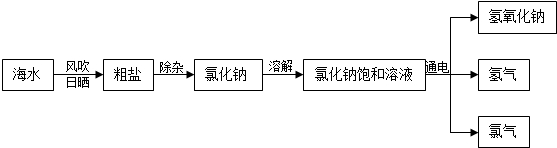
������һ�������Դ���⣬���ǿ��ԴӺ�ˮ����ȡ�Ȼ��ƣ������Ȼ��ƺ�ˮ��Ϊԭ����ȡ�������ơ�������������Cl2������1�����÷紵��ɹ���ԴӺ�ˮ����ȡ���Σ��ù�����Ҫ���� �����������ѧ�����仯��
��2���Ȼ��Ʊ�����Һ��ͨ�������·�����Ӧ�Ļ�ѧ����ʽ�� ��
��3��ij��ѧ��ȤС��Դ��ν��г����ᴿʵ�飮
�ٳ�ȡ5.0g���Σ���ҩ������10mlˮ�У�ֱ�����β����ܽ�Ϊֹ������Ҫ���е�ʵ����������˳��Ϊ������ʣ����Ρ� �����ţ���
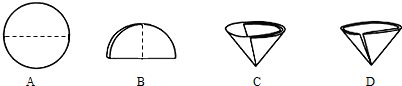
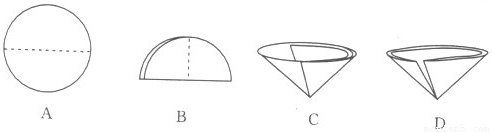
A���������� B��������� C������ D������
�ڹ��˲�������Ҫ��Բ����ֽ�۵���������ͼʾ�в��ó��ֵ������� �����ţ���
������ʱΪ�������ٹ���ɽ���������������ɲ�ȡ �ȴ�ʩ���õ��ľ����� ����������������
���ڴ����ᴿ��ʵ���У�ÿ�����趼��Ҫ�õ��IJ��������� ��
���𰸡���������1�����������仯�Ķ������
��2�����ݷ�Ӧ��������Ӧ��������д��ѧ����ʽ��ԭ����ȷ��д����ʽ
��3���ٸ��ݴ����ᴿ�IJ������
�ڸ��ݹ����������������
�۸���������������
�ܸ��ݴ����ᴿ���õ�����������
����⣺��1�����÷紵��ɹ���ԴӺ�ˮ����ȡ���Σ��ù�����Ҫ�����������仯��
��2����������Ϣ�Ȼ��ƺ�ˮ��Ϊԭ����ȡ�������ơ�������������Cl2�����ʻ�ѧ����ʽΪ2NaCl+2H2O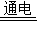 2NaOH+H2��+Cl2��
2NaOH+H2��+Cl2��
��3���ٴ��ξ��ܽ⡢���ˡ������������ε�������Ȼ���ټ�����ʣ��ʲ�������ΪDCAB
�ڽ�Բ����ֽ�۵����ó��ֵ������� D
������ʱΪ��ֹҺ��ɽ������ɵ���ʯ�������ȣ��õ��ľ����л����п����Ե����ʣ��ǻ���
���ܽ�������ò������ӿ��ܽ��ٶȣ�����ʱ����������ʱ���裬��ֹҺ��ֲ����ȣ����Һ�ηɽ�����˶�Ҫ�õ�������
�ʴ�Ϊ����1��������2��2NaCl+2H2O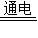 2NaOH+H2��+Cl2����3����DCAB
2NaOH+H2��+Cl2����3����DCAB
��D �۵���ʯ�������� �����ܲ�����
��������ȷ�����ᴿ�IJ��輰ע�������ǽ����ؼ���
��2�����ݷ�Ӧ��������Ӧ��������д��ѧ����ʽ��ԭ����ȷ��д����ʽ
��3���ٸ��ݴ����ᴿ�IJ������
�ڸ��ݹ����������������
�۸���������������
�ܸ��ݴ����ᴿ���õ�����������
����⣺��1�����÷紵��ɹ���ԴӺ�ˮ����ȡ���Σ��ù�����Ҫ�����������仯��
��2����������Ϣ�Ȼ��ƺ�ˮ��Ϊԭ����ȡ�������ơ�������������Cl2�����ʻ�ѧ����ʽΪ2NaCl+2H2O
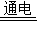 2NaOH+H2��+Cl2��
2NaOH+H2��+Cl2����3���ٴ��ξ��ܽ⡢���ˡ������������ε�������Ȼ���ټ�����ʣ��ʲ�������ΪDCAB
�ڽ�Բ����ֽ�۵����ó��ֵ������� D
������ʱΪ��ֹҺ��ɽ������ɵ���ʯ�������ȣ��õ��ľ����л����п����Ե����ʣ��ǻ���
���ܽ�������ò������ӿ��ܽ��ٶȣ�����ʱ����������ʱ���裬��ֹҺ��ֲ����ȣ����Һ�ηɽ�����˶�Ҫ�õ�������
�ʴ�Ϊ����1��������2��2NaCl+2H2O
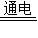 2NaOH+H2��+Cl2����3����DCAB
2NaOH+H2��+Cl2����3����DCAB��D �۵���ʯ�������� �����ܲ�����
��������ȷ�����ᴿ�IJ��輰ע�������ǽ����ؼ���

��ϰ��ϵ�д�
�����Ŀ