��Ŀ����
ij��ѧ��ȤС���ͬѧ�ɼ���һЩ����ʯ��Ʒ��Ϊ�ⶨ����Ʒ��̼��Ƶ�����������ȡ��Ʒ6g����ɷ�ĩ״�����ձ��У������м���10%��ϡ���Ტ���Ͻ��裬ǡ�ò��������ݲ���ʱ����֪���ʲ���ϡ���ᷴӦ��������ȥϡ����36.5g����1����ʵ�����ܷ���ϡ�������ϡ����______����ܡ����ܡ�����
��2������������Ϊ36%��Ũ����100g����������10%��ϡ����______g��
��3��������Ʒ��̼��Ƶ����������������������0.1%��
���𰸡���������1����ĩ״��̼��ƿ��Ժ�ϡ���ᷴӦ�����Ծݴ˽��
��2��������Һϡ��ǰ�����ʵ�������������ɽ��
��3���������ĵ�ϡ�����������Ϸ�Ӧ�Ļ�ѧ����ʽ���Լ����̼��Ƶ������������������Ʒ��̼��Ƶ�����������
����⣺��1����ĩ״��̼��ƿ��Ժ�ϡ���ᷴӦ����������ϡ����������ϡ���
��2����������10%��ϡ���������Ϊx�������ϡ��ǰ�����ʵ���������ɵã�
100g×36%=x?10%
��ã�x=360g
��3����6g��Ʒ��CaCO3������Ϊy
CaCO3+2HCl�TCaCl2+CO2��+H2O
100 73
y 36.5g×10%
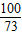 =
=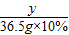
��ã�y=5g
��Ʒ��̼��Ƶ���������Ϊ��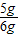 ×100%=83.3%
×100%=83.3%
����Ʒ��̼��Ƶ���������Ϊ83.3%��
�ʴ�Ϊ����1���ܣ�
��2��360��
��3��83.3%��
������Ҫ�����������Ŀ�����ȣ�Ҫ�������Ǹ��ݻ�ѧ��Ӧ����ʽ�ļ��㲽���ʽ���Լ���֮��ص�֪ʶ�ȣ�Ȼ���������������龰��ͼ����Ϣ�ȣ������ѧ�����֪ʶ�ͼ��ܣ�ϸ�µط������⣨��ͼ����Ϣ���ȸ�����Ϣ��Դ����ϸ�ĵ�̽��������������ĿҪ����������ѡ����ɣ�
��2��������Һϡ��ǰ�����ʵ�������������ɽ��
��3���������ĵ�ϡ�����������Ϸ�Ӧ�Ļ�ѧ����ʽ���Լ����̼��Ƶ������������������Ʒ��̼��Ƶ�����������
����⣺��1����ĩ״��̼��ƿ��Ժ�ϡ���ᷴӦ����������ϡ����������ϡ���
��2����������10%��ϡ���������Ϊx�������ϡ��ǰ�����ʵ���������ɵã�
100g×36%=x?10%
��ã�x=360g
��3����6g��Ʒ��CaCO3������Ϊy
CaCO3+2HCl�TCaCl2+CO2��+H2O
100 73
y 36.5g×10%
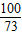 =
=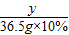
��ã�y=5g
��Ʒ��̼��Ƶ���������Ϊ��
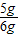 ×100%=83.3%
×100%=83.3%����Ʒ��̼��Ƶ���������Ϊ83.3%��
�ʴ�Ϊ����1���ܣ�
��2��360��
��3��83.3%��
������Ҫ�����������Ŀ�����ȣ�Ҫ�������Ǹ��ݻ�ѧ��Ӧ����ʽ�ļ��㲽���ʽ���Լ���֮��ص�֪ʶ�ȣ�Ȼ���������������龰��ͼ����Ϣ�ȣ������ѧ�����֪ʶ�ͼ��ܣ�ϸ�µط������⣨��ͼ����Ϣ���ȸ�����Ϣ��Դ����ϸ�ĵ�̽��������������ĿҪ����������ѡ����ɣ�

��ϰ��ϵ�д�
�����Ŀ
 12��̼�����ƣ�NaHCO3���׳�С�մ�����ʳƷ��ҽҩ��ҵ��ij��ѧ��ȤС���ͬѧ��̼�����Ƶ����ʽ���̽����
12��̼�����ƣ�NaHCO3���׳�С�մ�����ʳƷ��ҽҩ��ҵ��ij��ѧ��ȤС���ͬѧ��̼�����Ƶ����ʽ���̽����


 ��2012?��̨��ij��ѧ��ȤС���ͬѧ����ͼ��ʾװ�ý���ʵ�飨װ�����������ã����ȹر�ֹˮ�У�������������������Һ������ƿ�У�������յ�������̼���ٴ�ֹˮ�У�
��2012?��̨��ij��ѧ��ȤС���ͬѧ����ͼ��ʾװ�ý���ʵ�飨װ�����������ã����ȹر�ֹˮ�У�������������������Һ������ƿ�У�������յ�������̼���ٴ�ֹˮ�У� ij��ѧ��ȤС���ͬѧ��ʵ����������������Ϊ8%������������Һ��������ⶨijϡ���������ʵ�����������
ij��ѧ��ȤС���ͬѧ��ʵ����������������Ϊ8%������������Һ��������ⶨijϡ���������ʵ�����������