��Ŀ����
ij��ѧ��ȤС���ͬѧ��ʵ����������������Ϊ8%������������Һ��������ⶨijϡ���������ʵ�����������
��1������200g��������Ϊ8%������������Һ��
�ټ��㣺��Ҫ�������ƹ��������Ϊ?????? g��ˮ�����Ϊ?????? mL(ˮ���ܶȽ��ƿ���1g��cm3)��
�ڳ���������������ƽƽ�⣬Ȼ��??????????????? �����������Ⱥ�˳��ѡ����ĸ����ֱ����ƽƽ�⡣
A�����������ƹ�������ձ���??? B������Ҫ�������롢�ƶ�����
�ò��������ձ�������ֽ�����������Ƶ�ԭ����????????????????????? ��
���ܽ⣺����Ͳ��ȡ�����ˮ������ʢ���������ƹ�����ձ�����裬ʹ���ܽ⣬����ȴ�����¡�
�ܰ���õ���Һװ���Լ�ƿ��������Ƥ�������ϱ�ǩ��
��2����ͼ��ʾ���������Ƶ�����������Һ��20 gijϡ���ᷢ����Ӧ����Һ�¶ȵı仯�����
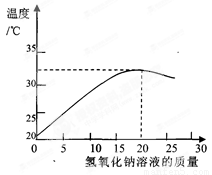
�ٸ��������жϣ�����������������Һ������Ϊ????????? ʱ����Ӧǡ����ȫ���С�
�ڵ�����15 g����������Һʱ��������Һ�е�����Ϊ?????????? ��д��ѧʽ����
���Լ���������ȫ��Ӧ��������Һ������������������д��������̣�????????????????? ��
��1����16?? 184??? �� B A? ���������׳��⣨���������ƾ��и�ʴ�ԣ�
��2����20g???? �� NaCl��HCl???? �� 5.85%
��������
�����������1������������������������ı���ʽ���Եó������ʵ�����=��Һ������������������������Ҫ�������ƹ��������=200g��8%=16g��ˮ������=200g-16g=184g���ڸ���ˮ���ܶ�1g��cm3������ˮ�����Ϊ=184mL���ڳ����̶�����������Ӧ�ȹ̶���������룬Ȼ��������ҩƷ�����Բ������Ⱥ�˳���ǣ�B A���������������׳��⣨���������ƾ��и�ʴ�ԣ������Գ����������ƹ����ʱ������ֽ��������Ӧ�÷��ڲ��������У��ձ���������
��2������кͷ�Ӧ��ų��������������ŷ�Ӧ�Ľ��У��������Ϸų�������Ӧֹͣʱ���ų����������ͼ����ʾ������������������Һ������Ϊ20g���¶���ߣ���ʾ��Ӧǡ����ȫ����
��3����Ϊ����������ǣ�����������������Һ������Ϊ20g����ʾ��Ӧǡ����ȫ���У����Ե�����15 g����������Һʱ����ʾϡ�����������������Һ�е����ʳ������ɵ�NaCl�����й�����HCl
?? ��4������������������Һ������Ϊ20g����������Ϊ8%����ʾ��Ӧǡ����ȫ���У������ĵ�������������=20g��8%=1.6g���ٸ��ݻ�ѧ����ʽ��NaOH+HCl=NaCl+H2O�������������NaCl������
�⣺����������NaCl������Ϊx
NaOH+HCl=NaCl+H2O
40??????? 58.5
1.6g??????? x
40��58.5 = 1.6g��x
x = 2.34g
�� ������Һ��������������=2.34g/20g+20g��100%=5.85%[
���㣺��Һ�����ƣ��кͷ�Ӧ�����ݻ�ѧ����ʽ���м���

 ��ĩ1�����ʽ���������ϵ�д�
��ĩ1�����ʽ���������ϵ�д� 12��̼�����ƣ�NaHCO3���׳�С�մ�����ʳƷ��ҽҩ��ҵ��ij��ѧ��ȤС���ͬѧ��̼�����Ƶ����ʽ���̽����
12��̼�����ƣ�NaHCO3���׳�С�մ�����ʳƷ��ҽҩ��ҵ��ij��ѧ��ȤС���ͬѧ��̼�����Ƶ����ʽ���̽����


 ��2012?��̨��ij��ѧ��ȤС���ͬѧ����ͼ��ʾװ�ý���ʵ�飨װ�����������ã����ȹر�ֹˮ�У�������������������Һ������ƿ�У�������յ�������̼���ٴ�ֹˮ�У�
��2012?��̨��ij��ѧ��ȤС���ͬѧ����ͼ��ʾװ�ý���ʵ�飨װ�����������ã����ȹر�ֹˮ�У�������������������Һ������ƿ�У�������յ�������̼���ٴ�ֹˮ�У� ij��ѧ��ȤС���ͬѧ��ʵ����������������Ϊ8%������������Һ��������ⶨijϡ���������ʵ�����������
ij��ѧ��ȤС���ͬѧ��ʵ����������������Ϊ8%������������Һ��������ⶨijϡ���������ʵ�����������