��Ŀ����
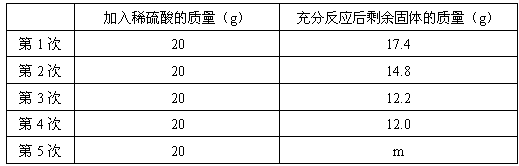
Cu��Zn�ĺϽ��Ϊ��ͭ���������ĵ����Ժ���ʴ�ԣ��������������������ij��ѧ��ȤС���ͬѧΪ�˲ⶨij��ͭ����ɣ�ȡ20g�û�ͭ��Ʒ���ձ��У������з�5�μ�����ͬ��������������ϡ���ᣬʹ֮��ַ�Ӧ��ÿ������ϡ�����������ʣ������������¼���±���
�Իش��������⣺
��1������������m��ֵΪ ��
��2����ͭ��Ʒ��п����������Ϊ ��
��3������ϡ��������������������Ƕ��٣�
��1������������m��ֵΪ ��
��2����ͭ��Ʒ��п����������Ϊ ��
��3������ϡ��������������������Ƕ��٣�
��1��12.0����12��
��2��40%
�⣺������ϡ�������������������Ϊx
Zn + H2SO4 === ZnSO4 + H2��
65 98
20g-17.4g=2.6g 20gx
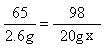 x=19.6%
x=19.6%
������ϡ���������ʵ���������Ϊ19.6%��
��2��40%
�⣺������ϡ�������������������Ϊx
Zn + H2SO4 === ZnSO4 + H2��
65 98
20g-17.4g=2.6g 20gx
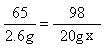 x=19.6%
x=19.6% ������ϡ���������ʵ���������Ϊ19.6%��

��ϰ��ϵ�д�
�����Ŀ
Cu��Zn�ĺϽ��Ϊ��ͭ���������ĵ����Ժ���ʴ�ԣ����������ֵ������������Ʒ��ij��ѧ��ȤС���ͬѧΪ�˲ⶨij��ͭ��Ʒ��п��������������150gϡ��������μ��뵽50g��ͭ��Ʒ�У�ÿ�γ�ַ�Ӧ�ⶨ����������������ʵ�����ݼ��±���
��ش��������⣺
��1��m= ��
��2���û�ͭ��Ʒ��п����������Ϊ ��
��3������ϡ���������ʵ���������Ϊ ��
| ��һ�� | �ڶ��� | ������ | |
| ����ϡ�����������g�� | 50 | 50 | 50 |
| ����������������g�� | 0.4 | m | 0.2 |
��1��m=
��2���û�ͭ��Ʒ��п����������Ϊ
��3������ϡ���������ʵ���������Ϊ
 ��2013?�ֿ���һģ��Cu��Zn�ĺϽ��Ϊ��ͭ���������ĵ����Ժ���ʴ�ԣ��������������������
��2013?�ֿ���һģ��Cu��Zn�ĺϽ��Ϊ��ͭ���������ĵ����Ժ���ʴ�ԣ��������������������