��Ŀ����
ʵ��������ʯ��ʯ��������ء�ϡ���ᡢ�������̡�����ľ�鼰����������
��ش��������⣺
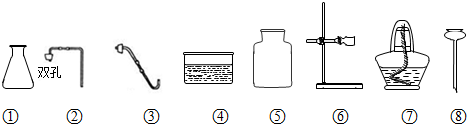
��1���������������______��
��2����������������ҩƷ��ȡ������̼����Ҫѡ���������______������ţ�����ȡ������Ļ�ѧ����ʽΪ______��
��3������ͬѧ����������һ֧�Թܣ�����������������ҩƷ��������ȡ����������ͼ�������ڻ��Ƶ���ȡ������װ��ͼ������������ǽ��䲹����������д��������Ӧ�Ļ�ѧ����ʽ��______��

��ش��������⣺
��1���������������______��

��2����������������ҩƷ��ȡ������̼����Ҫѡ���������______������ţ�����ȡ������Ļ�ѧ����ʽΪ______��
��3������ͬѧ����������һ֧�Թܣ�����������������ҩƷ��������ȡ����������ͼ�������ڻ��Ƶ���ȡ������װ��ͼ������������ǽ��䲹����������д��������Ӧ�Ļ�ѧ����ʽ��______��
����ʵ������ȡ������̼���õ�ҩƷ��ʯ��ʯ��ϡ���ᣬ���ڹ�Һ����Ҫ���ȵķ�Ӧ����˷���װ��Ӧѡ�٢ڢࣻ���������̼����ˮ���ܶȴ��ڿ���������ֻ��ѡ���������ſ������ռ��Ģݣ�
��ϡ����ʹ���ʯ��ʯ��ʯ��Ӧ������CO2����ƽ�ȣ������ֽ�Ϊ����������ѡ��ϡ������CO2����ʣ�
�ɸ������ȡ������ҩƷ�Ǹ�����غͶ������̣����ڹ̹̼��ȣ�����ȱ�ټ�����������Ҫ���ӵ������Թܣ���Ӧ�ķ���ʽ��2KMnO4
K2MnO4+MnO2+O2����
�ʴ�Ϊ��
��1������©��
��2���٢ڢݢࣻCaCO3+2HCl=CaCl3+H2O+CO2��
��3��2KMnO4
K2MnO4+MnO2+O2��
��ϡ����ʹ���ʯ��ʯ��ʯ��Ӧ������CO2����ƽ�ȣ������ֽ�Ϊ����������ѡ��ϡ������CO2����ʣ�
�ɸ������ȡ������ҩƷ�Ǹ�����غͶ������̣����ڹ̹̼��ȣ�����ȱ�ټ�����������Ҫ���ӵ������Թܣ���Ӧ�ķ���ʽ��2KMnO4
| ||
�ʴ�Ϊ��
��1������©��
��2���٢ڢݢࣻCaCO3+2HCl=CaCl3+H2O+CO2��
��3��2KMnO4
| ||

��ϰ��ϵ�д�
 �»����ܶ�Ա��ϵ�д�
�»����ܶ�Ա��ϵ�д� ����ͼ����ּ��������ҵ֣�ݴ�ѧ������ϵ�д�
����ͼ����ּ��������ҵ֣�ݴ�ѧ������ϵ�д�
�����Ŀ



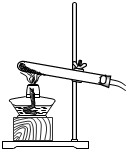
 ʵ���ҳ�����ȡ�����ķ����У�a����������غͶ������̷�ĩ�� b�����ȸ�����ع��壻c���ö������̷�ĩ������ʹ�������⣨H2O2����Һ�ֽ������������ʵ��װ����ͼ��ʾ��
ʵ���ҳ�����ȡ�����ķ����У�a����������غͶ������̷�ĩ�� b�����ȸ�����ع��壻c���ö������̷�ĩ������ʹ�������⣨H2O2����Һ�ֽ������������ʵ��װ����ͼ��ʾ�� ʵ���ҳ�����ȡ�����ķ����У�
ʵ���ҳ�����ȡ�����ķ����У�