��Ŀ����
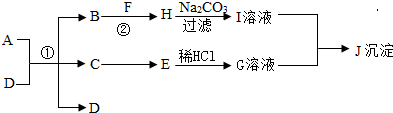
�������ʾ�Ϊ���л�ѧ�г��������ʣ�����A��B�����Ԫ����ͬ��B��D��E��FΪ�����D�Ǻ�ɫ���壬HΪ���������������õ����ʣ���ͼ������֮���ת����ϵ����ش�
��1��д���������ʵĻ�ѧʽ��F ��C
��2���õ���ҺI�IJ�������Ϊ ��
��3����GΪ��ɫ��Һ����E�Ļ�ѧʽΪ ��
��4����JΪ��ɫ������д���Ļ�ѧ����ʽ ��
��5������Ӧǰ��Ԫ�صĻ��ϼ۷����ı䣬������������ԭ��Ӧ�����жϷ�Ӧ�١��ڡ���������������ԭ��Ӧ���� ��
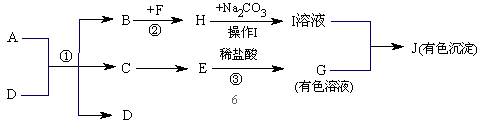
��2���õ���ҺI�IJ�������Ϊ ��
��3����GΪ��ɫ��Һ����E�Ļ�ѧʽΪ ��
��4����JΪ��ɫ������д���Ļ�ѧ����ʽ ��
��5������Ӧǰ��Ԫ�صĻ��ϼ۷����ı䣬������������ԭ��Ӧ�����жϷ�Ӧ�١��ڡ���������������ԭ��Ӧ���� ��
��1��CaO�� O2
��2������
��3��Fe2O3
��4��CuCl2+2NaOH===Cu(OH)2��+ 2NaCl
��5�� ��
��2������
��3��Fe2O3
��4��CuCl2+2NaOH===Cu(OH)2��+ 2NaCl
��5�� ��

��ϰ��ϵ�д�
�����Ŀ