��Ŀ����
��������AlN���������¡�������������Ժõ��������ܣ����㷺���ڴ��ģ���ɵ�·�������մɹ�ҵ������ҵ�ϴ��������л������X��X�к�������Ԫ�أ����ӿ����л�õ������ٽ�����X��N2��̼��һ�������·�Ӧ�Ƶ�AlN��CO����
��1���������մ����� ______��������ĸ��A����������B�����ǽ�������
��2��ͨ������£������е������������ԼΪ ______��������������������ȥ������̼��ˮ���Ŀ���Һ����ͨ�������¶Ƚ� ______�������������������Һ̬������������������
��3������X�Ļ�ѧʽ ______����AlN�Ļ�ѧ����ʽ ______��
��4�����ڵؿ��к���������Ԫ�صĵ� ______λ����Ԫ���������������к�����������һ��������������Ԫ������ĺ��������� ______��
��1���������մ����� ______��������ĸ��A����������B�����ǽ�������
��2��ͨ������£������е������������ԼΪ ______��������������������ȥ������̼��ˮ���Ŀ���Һ����ͨ�������¶Ƚ� ______�������������������Һ̬������������������
��3������X�Ļ�ѧʽ ______����AlN�Ļ�ѧ����ʽ ______��
��4�����ڵؿ��к���������Ԫ�صĵ� ______λ����Ԫ���������������к�����������һ��������������Ԫ������ĺ��������� ______��
��1���������մ��������ǽ������ϣ���ѡB��
��2����ͨ������£������е������������ԼΪ78%��
����Ϊ�������ܶȱ�����С����һ������Һ�������壬�ʵ�����Һ̬������������������
��3�����������غ㶨�ɺͻ�ѧ����ʽ��֪��X�Ļ�ѧʽ�к���Al�ĸ���Ϊ��2������N�ĸ���Ϊ��2-2=0������C�ĸ���Ϊ��3-3=0������O�ĸ���Ϊ��3����X�Ļ�ѧʽΪAl2O3����AlN�Ļ�ѧ����ʽΪ��Al2O3+N2+3C�T2AlN+3CO��
��4�����ڵؿ��к���������Ԫ�صĵ�3λ������������ԡ��ġ��Ρ����Ĺ��ܺ����߹��ܶ������������һ��������������Ԫ������ĺ��������飺�������Ʋ;��ٳ�������������װ�����ϣ�
�ʴ�Ϊ����1��B��
��2��78%��������
��3��Al2O3��Al2O3+N2+3C�T2AlN+3CO��
��4�� 3���������Ʋ;��ٳ�������������װ�����ϣ�
��2����ͨ������£������е������������ԼΪ78%��
����Ϊ�������ܶȱ�����С����һ������Һ�������壬�ʵ�����Һ̬������������������
��3�����������غ㶨�ɺͻ�ѧ����ʽ��֪��X�Ļ�ѧʽ�к���Al�ĸ���Ϊ��2������N�ĸ���Ϊ��2-2=0������C�ĸ���Ϊ��3-3=0������O�ĸ���Ϊ��3����X�Ļ�ѧʽΪAl2O3����AlN�Ļ�ѧ����ʽΪ��Al2O3+N2+3C�T2AlN+3CO��
��4�����ڵؿ��к���������Ԫ�صĵ�3λ������������ԡ��ġ��Ρ����Ĺ��ܺ����߹��ܶ������������һ��������������Ԫ������ĺ��������飺�������Ʋ;��ٳ�������������װ�����ϣ�
�ʴ�Ϊ����1��B��
��2��78%��������
��3��Al2O3��Al2O3+N2+3C�T2AlN+3CO��
��4�� 3���������Ʋ;��ٳ�������������װ�����ϣ�

��ϰ��ϵ�д�
 ѧ���쳵�����ּ��������ҵ�½����������ϵ�д�
ѧ���쳵�����ּ��������ҵ�½����������ϵ�д� �����ѧСѧ�꼶�νӵ������㽭��ѧ������ϵ�д�
�����ѧСѧ�꼶�νӵ������㽭��ѧ������ϵ�д� Сѧ�����ҵ���ϴ�ѧ������ϵ�д�
Сѧ�����ҵ���ϴ�ѧ������ϵ�д� ���Ž�����ٰθ��νӹ㶫���������ϵ�д�
���Ž�����ٰθ��νӹ㶫���������ϵ�д�
�����Ŀ
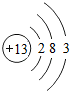 ����ԭ�Ӻ�������������������1������ԭ�ӵ�������Ϊ
����ԭ�Ӻ�������������������1������ԭ�ӵ�������Ϊ