��Ŀ����
ij��ѧ�о�С��Ե��ص�ʯ��ʯ��Ʒ�������о�����ȡʯ��ʯ��Ʒ8g����40gϡ������Ĵμ�����Ʒ�У����ʵ�����ݼ��±�������ʯ��ʯ��Ʒ�����ʲ�����ˮ���������ᷴӦ��������̼���ܽ⣩���Լ��㣺| ʵ����� | ��1�� | ��2�� | ��3�� | ��4�� |
| ����ϡ���������/g | 10 | 10 | 10 | 10 |
| ʣ��������ʵ�����/g | 5.5 | m | 1.6 | 1.6 |
��2��ʯ��ʯ��Ʒ��CaCO3 ������������
��3�������ʵ���й�����CO2��������
���𰸡����������ݱ�������ݿ�֪����һ�μ���10g��������ĵĹ���������8g-5.5g=2.5g����֪ÿ10g����������2.5g���壬�ɵ����μ��������ʣ��Ĺ���1.6g��ֻ������8g-2.5g-2.5g-1.6g=1.4g��1.6g��Ϊ���ʣ�̼����Ѿ�ȫ����Ӧ���ɴ˿�֪̼��Ƶ������������ʯ��ʯ��Ʒ��CaCO3 ��������������̼��Ƶ�����������̼���������ķ�Ӧ�����������̼��������
����⣺�ɱ�������ݿ�֪����һ�μ���10g��������ĵĹ���������8g-5.5g=2.5g����֪ÿ10g����������2.5g���壬�ɵ����μ��������ʣ��Ĺ�����1.6g��ֻ������8g-2.5g-2.5g-1.6g=1.4g��1.6g��Ϊ���ʣ�
��1���ɵ�һ�μ���10g����ʱ���ĵĹ���������8g-5.5g=2.5g�����Եڶ��μ���10g�������ĵ�̼�������ҲӦΪ2.5g������m=5.5g-2.5g=3g��
��2��ʯ��ʯ��Ʒ��CaCO3 �����������ǣ�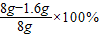 =80%��
=80%��
��3�����ʵ���й�����CO2������x
CaCO3+2HCl�TCaCl2+H2O+CO2��
100 44
8g-1.6g X
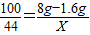 ��ã�X=2.816g
��ã�X=2.816g
�ʴ�Ϊ����1��3����2��ʯ��ʯ��Ʒ��CaCO3 ������������80%����3����ʵ���й�����CO2��������2.816g��
������������Ҫ�������û�ѧ����ʽ���м��������������Ĺؼ���ѧ���������ݣ��ҳ����ɽ��м��㣮
����⣺�ɱ�������ݿ�֪����һ�μ���10g��������ĵĹ���������8g-5.5g=2.5g����֪ÿ10g����������2.5g���壬�ɵ����μ��������ʣ��Ĺ�����1.6g��ֻ������8g-2.5g-2.5g-1.6g=1.4g��1.6g��Ϊ���ʣ�
��1���ɵ�һ�μ���10g����ʱ���ĵĹ���������8g-5.5g=2.5g�����Եڶ��μ���10g�������ĵ�̼�������ҲӦΪ2.5g������m=5.5g-2.5g=3g��
��2��ʯ��ʯ��Ʒ��CaCO3 �����������ǣ�
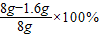 =80%��
=80%����3�����ʵ���й�����CO2������x
CaCO3+2HCl�TCaCl2+H2O+CO2��
100 44
8g-1.6g X
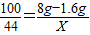 ��ã�X=2.816g
��ã�X=2.816g�ʴ�Ϊ����1��3����2��ʯ��ʯ��Ʒ��CaCO3 ������������80%����3����ʵ���й�����CO2��������2.816g��
������������Ҫ�������û�ѧ����ʽ���м��������������Ĺؼ���ѧ���������ݣ��ҳ����ɽ��м��㣮

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
ij��ѧ�о�С��Ե��ص�ʯ��ʯ��Ʒ�������о�����ȡʯ��ʯ��Ʒ8g����40gϡ������Ĵμ�����Ʒ�У����ʵ�����ݼ��±�������ʯ��ʯ��Ʒ�����ʲ�����ˮ���������ᷴӦ��������̼���ܽ⣩��
��1����4��ʣ�����ʵ�����m= g
��2��ʯ��ʯ��Ʒ��CaCO3����������Ϊ ��
��3������ϡ�������������������
| ʵ����� | ����ϡ���������/g | ʣ�����ʵ�����/g |
| ��1�� | 10 | 5.5 |
| ��2�� | 10 | 3 |
| ��3�� | 10 | 1.6 |
| ��4�� | 10 | m |
��2��ʯ��ʯ��Ʒ��CaCO3����������Ϊ
��3������ϡ�������������������
ij��ѧ�о�С��Ե��ص�ʯ��ʯ��Ʒ�������о�����ȡʯ��ʯ��Ʒ16g����80gϡ������Ĵμ�����Ʒ�У����ʵ�����ݼ��±�������ʯ��ʯ��Ʒ�����ʲ�����ˮ���������ᷴӦ��������̼���ܽ⣩��
��1����4��ʣ��������ʵ�����m= g
��2��ʯ��ʯ��Ʒ��CaCO3����������Ϊ ��
��3������ϡ�������������������
| ʵ����� | ����ϡ���������/g | ʣ��������ʵ�����/g |
| ��1�� | 20 | 11 |
| ��2�� | 20 | 6 |
| ��3�� | 20 | 3.2 |
| ��4�� | 20 | m |
��2��ʯ��ʯ��Ʒ��CaCO3����������Ϊ
��3������ϡ�������������������