��Ŀ����
18��Ϊ�Ƚ�Fe3+��Cu2+��H2O2�ֽⷴӦ�Ĵ�Ч������������ͬѧ�ֱ��������ͼ1��ͼ2��ʾ��ʵ�飮���������в���ȷ���ǣ�������

������A�����ݷ�Ӧ�ľ��ҳ̶���ʵ������Ĺ�ϵ�жϣ�
B�����ݴ����Ķ�������ü����Ʊ���������жϣ�
C�����ݷ�Ӧ���ʵĺ����жϣ�
D�����������Լ���ԭ���жϣ�
B�����ݴ����Ķ�������ü����Ʊ���������жϣ�
C�����ݷ�Ӧ���ʵĺ����жϣ�
D�����������Լ���ԭ���жϣ�
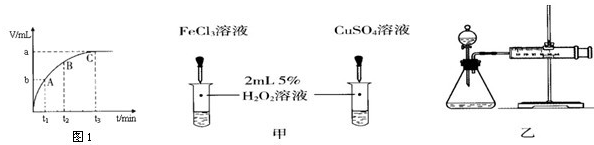
����⣺A����Ӧ�ľ��ҳ̶���ʵ�������йأ���ӦԽ���ң��������������Խ�죬��������Խ�죬������ȷ��
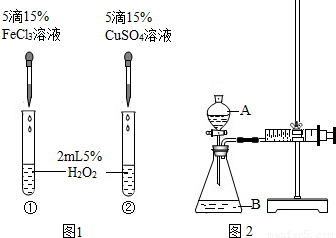
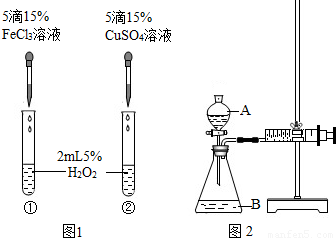
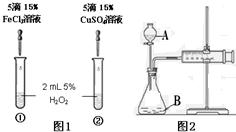
B�������ܸı仯ѧ��Ӧ�����ʣ������Ǽӿ����ʣ���ͼ1��ʾʵ���з�Ӧ����Ϊ�٣��ڣ���һ��˵���Ȼ���������ͭ��Ч���ã�����������������Ӳ�ͬ��Ҫ֤��Fe3+��Cu2+��H2O2�ֽ��Ч���ã���Ҫʹѡ����Լ���������������ͬ�����Դ���
C����Ӧ���ʿ��õ�λʱ���ڲ��������������ʾ����װ�ÿ�ͨ��ע����������λ�ñ仯����������������������ȷ��
D�����װ�õ�������һ����������ѹ��ԭ������ͼ2װ���У��ر�A����������ע�����Ļ�������һ�����룬��һ��ʱ������ɿ�������������ص�ԭλ��˵�����������ã�����©����������ȷ��
��ѡB��
B�������ܸı仯ѧ��Ӧ�����ʣ������Ǽӿ����ʣ���ͼ1��ʾʵ���з�Ӧ����Ϊ�٣��ڣ���һ��˵���Ȼ���������ͭ��Ч���ã�����������������Ӳ�ͬ��Ҫ֤��Fe3+��Cu2+��H2O2�ֽ��Ч���ã���Ҫʹѡ����Լ���������������ͬ�����Դ���
C����Ӧ���ʿ��õ�λʱ���ڲ��������������ʾ����װ�ÿ�ͨ��ע����������λ�ñ仯����������������������ȷ��
D�����װ�õ�������һ����������ѹ��ԭ������ͼ2װ���У��ر�A����������ע�����Ļ�������һ�����룬��һ��ʱ������ɿ�������������ص�ԭλ��˵�����������ã�����©����������ȷ��
��ѡB��
�����������ʹ������dz��л�ѧ�н��������������������������մ�����һ�䣬�����䡱���ص��ǽ���Ĺؼ���

��ϰ��ϵ�д�
 ��ǰ����ϵ�д�
��ǰ����ϵ�д�
�����Ŀ
Ϊ���о���������Թ�������ֽ����ʵ�Ӱ�죬ijͬѧ��������ʵ�飬��ش�
| ��� | ���� | ʵ������ |
| �� | �ֱ����Թ�A��B�м���5mL 5% H2O2��Һ��������2�ε�Ũ�� FeCl3��Һ�����Թ��о����������ݳ���ʱ�����Թ�A����ʢ��5��������ˮ���ձ��н��ݣ����Թ�B����ʢ��40��������ˮ���ձ��н��ݣ� | �Թ�A�в��ٲ������ݣ��Թ�B�в��������������� |
| �� | ��ȡ��֧�Թֱܷ����5mL 5% H2O2��Һ��5mL 10% H2O2��Һ | �Թ�A��B�о�δ���Լ��������ݲ����� |
��2��ʵ��ٵ�Ŀ����______��
ʵ���еμ�FeCl3��Һ��Ŀ����______��
��3��ʵ���δ�۲쵽Ԥ�ڵ�ʵ������Ϊ�˰�����ͬѧ�ﵽʵ��Ŀ�ģ�������Ķ����������ĸĽ������______����ʵ�����ṩ���Լ�����
��4��ijͬѧ��50mLһ��Ũ�ȵ�H2O2��Һ�м���һ�����Ķ������̣��ų����������뷴Ӧʱ��Ĺ�ϵ��ͼ1��ʾ����A��B��C��������ʾ��˲ʱ��Ӧ������������______��
��5�������ǻ�ѧ��Ӧǰ��______ ��δ�ı��һ�ѧ���ʣ�
��6������H2O2�ֽⷴӦ��Cu2+Ҳ��һ���Ĵ����ã�Ϊ�Ƚ�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч����ij��ѧ�о�С���ͬѧ�ֱ��������ͼ�ס�����ʾ��ʵ�飮��ش�������⣺

�ٶ��Է�������ͼ��ͨ���۲�______�����ԱȽϵó����ۣ���ͬѧ�����FeCl3��Ϊ______��Ϊ��������������______��
�ڶ�����������ͼ����ʾװ��������ʵ�飬ʵ��ʱ��������40mL����Ϊ����������Ӱ��ʵ������ؾ��Ѻ��ԣ�ʵ������Ҫ������������______��