��Ŀ����
ijУ��ѧ��ȤС��Ϊ��ȷ�ⶨijʯ����̼��Ƶ����������������Ƚ�ʯ�Ϸ��飬Ȼ���ȡ3�ݣ��ֱ����뵽3��ϡ�����У�ʯ���е����ʲ��μӷ�Ӧ��������Ӧֹͣ���������ݼ�¼���±���| 1 | 2 | 3 | |
| ʯ������/g | 50 | 50 | 100 |
| ϡ��������/g | 240 | 300 | 200 |
| CO2��������/g | 17.6 | 17.6 | 17.6 |
��1��50gʯ��ǡ����ȫ��Ӧʱ������ϡ����������� g��
��2����ʯ����̼��Ƶ�����������
��3��ǡ����ȫ��Ӧʱ������Һ����Һ���������������������ȷ��0.1%����
���𰸡���������1�����ݵ�1��ʵ��͵�3��ʵ����бȽϽ��з�������3�����ݶ�����̼�����������̼��Ƶ�����������̼��Ƶ�����������Ʒ���������ɣ�
����⣺��1����1�κ͵�2��ʵ��ȽϿ�֪50gʯ����ȫ��Ӧֻ������17.6g������̼���ٸ��ݵ�1��ʵ��͵�3��ʵ����бȽϣ���֪50gʯ����ȫ��Ӧ����17.6g������̼�����Ե�4���ǰ���100gϡ���ᷴӦ��ȡ17.6g������̼������50gʯ��ǡ����ȫ��Ӧʱ������ϡ�����������200g��
��2���⣺��ʯ����CaCO3������Ϊx�����ɵ��Ȼ���������y��
CaCO3+2HCl=CaCl2+CO2��+H2O
100 111 44
x y 17.6g
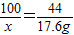 =
=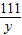
��ã�x=40g y=44.4g
��CaCO3����������=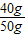 ×100%=80.0%
×100%=80.0%
ǡ�÷�Ӧʱ��Ҫ�������������200g���ʴ�ʱ��Һ��������������Ϊ��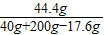 ×100%��20.0%
×100%��20.0%
�ʴ�Ϊ����1��200g����3����ʯ����CaCO3����������Ϊ80.0%����3����һ�ݷ�Ӧ��������Һ����Һ��������������ԼΪ20.0%��
�����������ǶԻ�ѧ����ʽ������ۺϿ����⣬���������غ㶨�ɣ���Ӧ��������Һ������=̼��Ƶ�����+����ϡ�����������-�ų�������̼����������ǽ���Ĺؼ���
����⣺��1����1�κ͵�2��ʵ��ȽϿ�֪50gʯ����ȫ��Ӧֻ������17.6g������̼���ٸ��ݵ�1��ʵ��͵�3��ʵ����бȽϣ���֪50gʯ����ȫ��Ӧ����17.6g������̼�����Ե�4���ǰ���100gϡ���ᷴӦ��ȡ17.6g������̼������50gʯ��ǡ����ȫ��Ӧʱ������ϡ�����������200g��
��2���⣺��ʯ����CaCO3������Ϊx�����ɵ��Ȼ���������y��
CaCO3+2HCl=CaCl2+CO2��+H2O
100 111 44
x y 17.6g
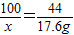 =
=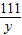
��ã�x=40g y=44.4g
��CaCO3����������=
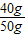 ×100%=80.0%
×100%=80.0%ǡ�÷�Ӧʱ��Ҫ�������������200g���ʴ�ʱ��Һ��������������Ϊ��
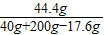 ×100%��20.0%
×100%��20.0%�ʴ�Ϊ����1��200g����3����ʯ����CaCO3����������Ϊ80.0%����3����һ�ݷ�Ӧ��������Һ����Һ��������������ԼΪ20.0%��
�����������ǶԻ�ѧ����ʽ������ۺϿ����⣬���������غ㶨�ɣ���Ӧ��������Һ������=̼��Ƶ�����+����ϡ�����������-�ų�������̼����������ǽ���Ĺؼ���

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
 ijУ��ѧ��ȤС��Ϊ�˲ⶨijһ̼������Ʒ��̼���Ƶ�������������������ʵ�飬ȡ13.25g̼������Ʒ�����ʼȲ�������Ҳ������ˮ�������ձ��У�����95.8gˮʹ����ȫ�ܽ⣬�������м���ϡ���ᣬ��÷�Ӧ�ų��������������������ϡ�����������ϵ��������ͼ��ʾ����������������⣺
ijУ��ѧ��ȤС��Ϊ�˲ⶨijһ̼������Ʒ��̼���Ƶ�������������������ʵ�飬ȡ13.25g̼������Ʒ�����ʼȲ�������Ҳ������ˮ�������ձ��У�����95.8gˮʹ����ȫ�ܽ⣬�������м���ϡ���ᣬ��÷�Ӧ�ų��������������������ϡ�����������ϵ��������ͼ��ʾ����������������⣺






