��Ŀ����
ij��ѧ��ȤС�������Һ�����ƺʹ��εij����ᴿʵ�飮��1������200g��������Ϊ8%������������Һ��
�ټ��㣺��Ҫ�������ƹ��������Ϊ �ˣ���Ҫˮ�����Ϊ mL��ˮ���ܶȽ��ƿ���1g/mL����
�ڳ���������������ƽƽ�⣬��һ���ձ�����������ƽ�� �̣�������������Ȼ�� �����������Ⱥ�˳��ѡ����ĸ����ֱ����ƽƽ�⣮
A�����������ƹ�������ձ��� B������Ҫ�������롢�ƶ�����
�ò��������ձ�������ֽ�����������Ƶ�ԭ���� ��
���� ��ȡ����Ҫ��ˮ������ʢ���������ƹ�����ձ��У��� ���裬ʹ���ܽ⣬����ȴ�����£�
�ܽ����ƺõ���Һ�����Լ�ƿ������������ ���ŵ�ָ���ĵط���
��2�����εij����ᴿ��
�ٳ�ȡ5.0�˴��Σ���ҩ�����뵽10mLˮ�У�ֱ�����β����ܽ�Ϊֹ��������е�ʵ����������˳��Ϊ������ʣ����Ρ� �����ţ���
A������ B��������� C������ D����������
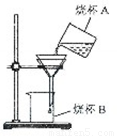
�ڹ��˲�����ͼ��ʾ��ָ�����еĴ���֮���� �� �� ��
�����������к�ǿ�ĸ�ʴ�ԣ�����ʱӦ���ڲ��������г��������Ӧ�ȳƳ��ձ����������ټ���������������Ƶij�����
����ȡҺ��һ������Ͳ����ȡ��Ϊ�˼ӿ��������Ƶ��ܽ⣬Ӧʹ�ò��������н��裬���Ծݴ�����
��������ϵ���ҺӦ���ϱ�ǩ��ţ����Ծݴ�����
��2����Ϊ��������ᴿ���ò��ʣ��ڶԴ����ܽ⡢���ˡ���������Ҫ���������ε�������Ȼ��������ʳ���������ܽ���ε������ȣ�������ʣ�
�ڸ��ݹ��˲�����Ҫ��һ���������������۲�ͼʾ�IJ�����ָ�������д��ڵĴ���
����⣺��1��������20��������Ϊ8����������Һ����Ҫ�������Ƶ�����=20×8=16����Ҫˮ������=200g-16g=184g����ˮ���ܶ�Ϊ1g/mL��������ˮ�����Ϊ184mL��
��Ϊ��ֹ�������Ƹ�ʴ��ƽ���̣�ֽ���ױ���ʴ�����Գ���ʱӦ���������Ʒ����ձ��н��г������ȳƳ��ձ���������Ȼ�������������룬���������˳�������������������Ƶ�������
��Ȼ������Ͳ����ȡ184mLˮ������ʢ���������Ƶ��ձ��У��ò������裬���ӿ��������Ƶ��ܽ⣬��ȫ���ܽ���ȴ���������������Һ��
��Ϊ���ܹ������ʶ�����Һ��������ɺ�Ӧ���ϱ�ǩ���棻
��2���ٴ��ε��ᴿ����˳��Ϊ�ܽ⡢���ˡ�������Ϊ������ʻ���Ҫ�Ƴ����ܽ���ε��������Ƴ�����������ʳ�ε������������м��㣻���˳��ΪACDB��
��ͼʾ�Ĺ��˲������ڶദ����δ�ò��������������ڵ���Һ��ʱʹ��ֽ������ֽ�߳�©����Ե���ᵼ��Һ������©������ֽδ����©���ڱڣ�Һ�����ֽ��©��֮�����������˲����ף�©���¶˼���δ�����ձ�B���ڱڣ�Һ������С�һὦ��
�ʴ�Ϊ����1����16��184��
����BA���������ƾ��и�ʴ�ԣ�
����Ͳ����������
�����ϱ�ǩ��
��2����ACDB��
��δ�ò����������� ��ֽ�߳�©����Ե����ֽδ����©���ڱڣ� ©������ĩ��δ�����ձ�B���ڱڣ�����˳��Ҫ�������ɣ���
������ע����˲���Ҫ��һ����������������һ������ֽ����©���ڱڣ����ͣ���ֽ����©����Һ�������ֽ���أ�������������ĩ��Ҫ����ֽ���㴦����ձ���Ҫ�벣�����в����©���¶˿�Ҫ���ձ��ڱ����

 ��У����ϵ�д�
��У����ϵ�д�ij��ɫ�����������������(��ѧʽΪBa(OH)2)�����������е�һ�ֻ�������ɡ�Ϊȷ���ù������ʵijɷ֣�ij![]() ��ѧ��ȤС���������̽��������������Ķ��й���Ϣ���ش�������⡣
��ѧ��ȤС���������̽��������������Ķ��й���Ϣ���ش�������⡣
���������ϡ�(1) Ba(OH)2��һ��ǿ�����NaOH���ƵĻ�ѧ���ʡ�
(2)�����ᡢ��ε��ܽ������±���ʾ��
|
| OH— | Cl— | SO42— | CO32— |
| Na+ | �� | �� | �� | �� |
| Ba2+ | �� | | ���� | ���� |
��������롿����I ���ù�����NaOH
����ù�����Ba(OH)2
����ù�����Ba(OH)2��NaOH�Ļ����
�����ʵ�顿
| ʵ�鲽�� | ʵ������ | ʵ����� |
| ��ȡ���� | �а�ɫ�������� | ����ԭ��Ʒ��һ������ ������ѧʽ) |
| ������м����μ���������Һ��ֱ��������������Ϊֹ�����ú���pH��ֽ�����Һ����� | ��Һ��ǿ���� | ����ԭ��Ʒ�л������������� |
���������ۡ�
(1)ʵ�鲽����з�����Ӧ�Ļ�ѧ����ʽΪ ![]() ��
��
(2)ʵ�鲽��ڵó���ʵ����� (���ȷ������ȷ��)��
(3)���������ʵ������ϡ���������������Һ��ʵ�鲽���ʵ���������ͬ��������жϳ����� �dz����ġ�
 ��2013?����һģ��ij��ѧ��ȤС���������ͼ��ʾ����Ȫʵ�飬����ͷ�ι��е�ˮ������ƿ��Ȼ���ֹˮ�У�����ƿ�ڳ��������ĺ�ɫ��Ȫ��
��2013?����һģ��ij��ѧ��ȤС���������ͼ��ʾ����Ȫʵ�飬����ͷ�ι��е�ˮ������ƿ��Ȼ���ֹˮ�У�����ƿ�ڳ��������ĺ�ɫ��Ȫ��