��Ŀ����
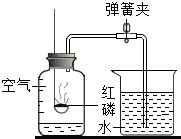 ʹ����ͼ��ʾװ�òⶨ�����������ĺ�������ش�
ʹ����ͼ��ʾװ�òⶨ�����������ĺ�������ش���1��Ϊȷ��ʵ��ɹ���ʵ��ǰһ��Ҫ����װ�õ�
������
������
��2����
ȼ�ճ�
ȼ�ճ�
����װ�����ƣ������Ӻ�����Ӧ������
����
������⡱����������������������3��д������ȼ��ʱ�Ļ�ѧ����ʽ
4P+5O2
2P2O5
| ||
4P+5O2
2P2O5
����ʱ���Թ۲쵽��������
| ||
�����ƹ⣬�ų�������ð�������İ���
�����ƹ⣬�ų�������ð�������İ���
��ȼ��ֹͣ������ȴ����ɼУ���4����ʵ�����յõ��Ľ�����
����������Լռ������������֮һ
����������Լռ������������֮һ
�����������ݲⶨ�����������ĺ����IJ��衢������ѹǿ��֪ʶ�������������©���ͺ��������㵼�µĽ�����ǣ����ݺ���ȼ�յ��������������������ѹǿ��С���µ������ǣ�����ʵ��������ʵ����ۣ�
����⣺��1�����װ��©������ʹװ������ѹǿ��ȣ�ˮ���ᱻѹ��ƿ�ڣ�
��2�����������㲻�ܰ�װ�������������꣬���½����ˮƫ�٣���ó�������ۣ�
��3��д������ȼ��ʱ�Ļ�ѧ����ʽ 4P+5O2
2P2O5����ʱ���Թ۲쵽�������� �����ƹ⣬�ų�������ð�������İ��̣�
��4�������ˮ��������DZ����ĵ����������������˵������������Լռ������������֮һ��
�ʴ�Ϊ����1�������ԣ���2��ȼ�ճף���������3��4P+5O2
2P2O5
�����ƹ⣬�ų�������ð�������İ��̣���4������������Լռ������������֮һ��
��2�����������㲻�ܰ�װ�������������꣬���½����ˮƫ�٣���ó�������ۣ�
��3��д������ȼ��ʱ�Ļ�ѧ����ʽ 4P+5O2
| ||
��4�������ˮ��������DZ����ĵ����������������˵������������Լռ������������֮һ��
�ʴ�Ϊ����1�������ԣ���2��ȼ�ճף���������3��4P+5O2
| ||
�����ƹ⣬�ų�������ð�������İ��̣���4������������Լռ������������֮һ��
�����������ؼ���Ҫ֪��װ�ñ����ܷ⣬Ϊ�˰����������꣬�����������ĺ��ף�֪������ˮ������������ĵ������������

��ϰ��ϵ�д�
�����Ŀ
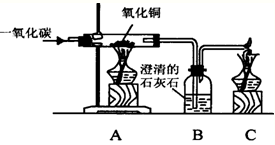 ijͬѧʹ����ͼ��ʾװ�ã�����һ����̼������ͭ�ķ�Ӧ����ش������й����⣺
ijͬѧʹ����ͼ��ʾװ�ã�����һ����̼������ͭ�ķ�Ӧ����ش������й����⣺ ij��ѧС��������ͼ��ʾװ�òⶨͭп�Ͻ���Ʒ��п������������ͼ�й̶�װ������ȥ��̽���������£�
ij��ѧС��������ͼ��ʾװ�òⶨͭп�Ͻ���Ʒ��п������������ͼ�й̶�װ������ȥ��̽���������£�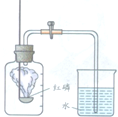 ijͬѧ������ͼ��ʾװ�òⶨ�����������ĺ��������õ��ɼм�ס��Ƥ�ܣ���ȼ���ף�����ƿ�в�����ƿ�����Ⱥ���ȼ��Ϩ����ٴ��ɼУ����ּ���ƿ�е�ˮ�������ݻ���
ijͬѧ������ͼ��ʾװ�òⶨ�����������ĺ��������õ��ɼм�ס��Ƥ�ܣ���ȼ���ף�����ƿ�в�����ƿ�����Ⱥ���ȼ��Ϩ����ٴ��ɼУ����ּ���ƿ�е�ˮ�������ݻ���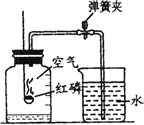 ʹ����ͼ��ʾװ�òⶨ�����������ĺ�������ش�
ʹ����ͼ��ʾװ�òⶨ�����������ĺ�������ش�