��Ŀ����
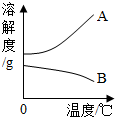
�±���NaOH��Ca��OH��2���ܽ�����ݣ���ش��������⣮| �¶ȣ��棩 | 20 | 40 | 60 | 80 | 100 | ||
| �ܽ�� ��g�� | NaOH | 31 | 91 | 111 | 129 | 313 | 336 |
| Ca��OH��2 | 0.19 | 0.17 | 0.14 | 0.12 | 0.09 | 0.08 | |
��2����80��ʱNaOH�ı�����Һ������20�棬���Կ�����������______������20��ʱCa��OH��2�ı�����Һ������Һ���������м���һ����CaO��õ�����Һ������Һ������ʱ��Һ����������������______�ף��������=����
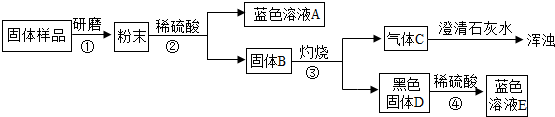
��3��ij��ȤС��Բ��ֱ��ʵ��������ƹ�������ᴿ����������²������̣���ش�

������ڷ�Ӧ�Ļ�ѧ����ʽΪ______���������Ca��OH��2��Ŀ����______��
������ҺB�е�������______��______��д��ѧʽ����������������ľ�������Ǽ���Ũ����______�����ˣ�
���𰸡���������1�����ñ��������������������Ƶ��ܽ����ֵ���ҵ���ص���Ϣ��
��2�������������Ƶ��ܽ�����¶Ƚ��Ͷ���С��֪ʶ�Լ����������ܽ�����¶����߶���С��ͬʱ�����ʯ������ˮ���ȵ�֪ʶ������⣮
��3��I�����ʵ��������Ƴ��Ậ��̼���ƣ�̼�������������Ʒ�Ӧ������̼������������ƣ��������������ƻ��̼��������ĸ��ɾ�һЩ��
II����ҺB�лẬ��ʣ����������Ƽ����������������ʣ����������ƺ��������Ƶ��ܽ�����¶ȵĹ�ϵ��ȷ���������ǵķ�ʽ��
����⣺��1����ͬ�¶��������������������Ƶ��ܽ�ȴ�С����ܴ�
�ʴ�Ϊ���������Ƶ��ܽ��ԶԶ�����������Ƶ��ܽ�ȣ�
��2�������������Ƶ��ܽ�����¶Ƚ��Ͷ���С���ʽ��»�ʹ��Һ�������ʣ���������Һˮ�������Һ�¶����ߣ����������ܽ�����¶����߶���С���ʿ��γɸ���ʱ�ı�����Һ������ҺҪ�ȵ���ʱ������������С��
�ʴ�Ϊ����Һ�����ǣ���
��3��I�����ʵ��������Ƴ��������������������̼��Ӧ����̼���ƣ�̼�������������Ʒ�Ӧ������̼������������ƣ�Ϊ��̼��������ĸ��ɾ�һЩ�������������������ƣ�
�ʴ�Ϊ��Na2CO3+Ca��OH��2=CaCO3��+2NaOH������Һ�е�̼������ȫת��Ϊ�������ƣ�
II��������Һ�м����˹������������ƣ�������Һ�лẬ�������������������ƣ���Ϊ���������ʵ��ܽ�����¶ȵı仯����ܴʿɲ��ý��½ᾧ�ķ�ʽ���з��룮
�ʴ�Ϊ��Ca��OH��2��NaOH�����½ᾧ
�����������Ƕ��ܽ��֪ʶ������������й�֪ʶ���飬����Ĺؼ��Ƕ��������е���Ϣ����������ܽ��֪ʶ�Ի����ķ������̽�֣��������Ի����������Ŀ��
��2�������������Ƶ��ܽ�����¶Ƚ��Ͷ���С��֪ʶ�Լ����������ܽ�����¶����߶���С��ͬʱ�����ʯ������ˮ���ȵ�֪ʶ������⣮
��3��I�����ʵ��������Ƴ��Ậ��̼���ƣ�̼�������������Ʒ�Ӧ������̼������������ƣ��������������ƻ��̼��������ĸ��ɾ�һЩ��
II����ҺB�лẬ��ʣ����������Ƽ����������������ʣ����������ƺ��������Ƶ��ܽ�����¶ȵĹ�ϵ��ȷ���������ǵķ�ʽ��
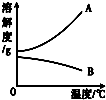
����⣺��1����ͬ�¶��������������������Ƶ��ܽ�ȴ�С����ܴ�
�ʴ�Ϊ���������Ƶ��ܽ��ԶԶ�����������Ƶ��ܽ�ȣ�
��2�������������Ƶ��ܽ�����¶Ƚ��Ͷ���С���ʽ��»�ʹ��Һ�������ʣ���������Һˮ�������Һ�¶����ߣ����������ܽ�����¶����߶���С���ʿ��γɸ���ʱ�ı�����Һ������ҺҪ�ȵ���ʱ������������С��
�ʴ�Ϊ����Һ�����ǣ���
��3��I�����ʵ��������Ƴ��������������������̼��Ӧ����̼���ƣ�̼�������������Ʒ�Ӧ������̼������������ƣ�Ϊ��̼��������ĸ��ɾ�һЩ�������������������ƣ�
�ʴ�Ϊ��Na2CO3+Ca��OH��2=CaCO3��+2NaOH������Һ�е�̼������ȫת��Ϊ�������ƣ�
II��������Һ�м����˹������������ƣ�������Һ�лẬ�������������������ƣ���Ϊ���������ʵ��ܽ�����¶ȵı仯����ܴʿɲ��ý��½ᾧ�ķ�ʽ���з��룮
�ʴ�Ϊ��Ca��OH��2��NaOH�����½ᾧ
�����������Ƕ��ܽ��֪ʶ������������й�֪ʶ���飬����Ĺؼ��Ƕ��������е���Ϣ����������ܽ��֪ʶ�Ի����ķ������̽�֣��������Ի����������Ŀ��

��ϰ��ϵ�д�
�����Ŀ
�±���NaOH��Ca��OH��2���ܽ�����ݣ���ش��������⣮
| �¶ȣ��棩 | 0 | 20 | 40 | 60 | 80 | 100 | |
| �ܽ�� ��g�� | NaOH | 31 | 91 | 111 | 129 | 313 | 336 |
| Ca��OH��2 | 0.19 | 0.17 | 0.14 | 0.12 | 0.09 | 0.08 | |
��2��ij��ȤС��Բ��ֱ��ʵ��������ƹ�������ᴿ���������ͼ�������̣���ش�

�ٲ���ڷ�Ӧ�Ļ�ѧ����ʽΪ________��
����ҺB�е����������֣����Ƿֱ���________��д��ѧʽ����
�۲�����������ľ�������Ǽ���Ũ����________�����ˣ�