��Ŀ����
�ձ���װ��һ�������������ͭ�Ļ����Һ����֪����Һ�к�H2SO4������Ϊ9.8 g��ijͬѧΪ�ⶨ�û����Һ������ͭ�����������ձ�������10%��NaOH��Һ���õ�������������¼���£�

(1)�õ�������������Ϊ___________g���û����Һ������ͭ������Ϊ___________g��
(2)�μӷ�Ӧ��NaOH��Һ���������Ƕ��ٿ�?(Ҫ��д���������)
(3)�����ڸû����Һ�м���NaOH��Һ���������ɳ��������仯��ϵ�����ߡ�
(1)9.8 16.0
(2)�⣺�������ᷴӦ��NaOH������Ϊx����CuSO4��Ӧ��NaOH������Ϊy��
H2SO4+2NaOH===Na2SO4+2H2O CuSO4+2NaOH===Cu(OH)2![]() +Na2SO4
+Na2SO4
98 80 80 98
9.8 g x y 9.8 g
![]()
![]()
x=8 g y=8 g
�ʲμӷ�Ӧ��NaOH��Һ��������Ϊ![]()
�����������вμӷ�Ӧ��NaOH��Һ��������Ϊ160�ˡ�
 (3)(ץס��㡢�۵㼰�������Ƹ���)
(3)(ץס��㡢�۵㼰�������Ƹ���)

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| ����NaOH��Һ������/g | 50.0 | 100.0 | 150.0 | 200.0 | 250.0 |
| ���ɳ���������/g | 0.0 | 2.5 | 8.6 | 9.8 | 9.8 |
��2���μӷ�Ӧ��NaOH��Һ���������Ƕ��ٿˣ���Ҫ��д��������̣�
��3�������ڸû����Һ�м���NaOH��Һ���������ɳ��������仯��ϵ�����ߣ����������
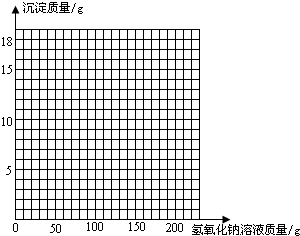
| ����NaOH��Һ������/g | 50.0 | 100.0 | 150.0 | 200.0 | 250.0 |
| ���ɳ���������/g | 0.0 | 2.45 | 8.6 | 9.8 | 9.8 |
��2���μӷ�Ӧ��NaOH��Һ���������Ƕ��ٿˣ���Ҫ��д��������̣�