��Ŀ����
�ϳɰ��������ѧ�����ϵ�һ���ش�ͻ�ƣ�����ᷢչ����������˾��ף��ϳɰ��Ĺ�������ͼ���£�
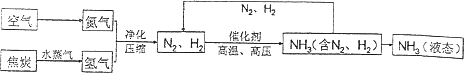
(1)�ϳɰ���Ҫ�ĵ������Կ����������е������������ԼΪ________��
(2)��ȡ�����ķ�Ӧ����Ϊ��C��H2O![]() CO��H2��CO��H2O
CO��H2��CO��H2O![]() CO2��H2������������ѧ����ʽ�б��������Ե�������________(�ѧʽ)��
CO2��H2������������ѧ����ʽ�б��������Ե�������________(�ѧʽ)��
(3)ԭ����N2��H2�г�����CO2��ˮ��������ӦǰҪ�����������Ĺ�����________��
(4)д��N2��H2��Ӧ����NH3�Ļ�ѧ����ʽ________��
(5)���е㲻ͬ��������뿪����������Һ�����뷨���磬�����¶��ڣ�183��ʱ���ɽ�������N2��O2���룮�����±������ʵķе��жϣ�Ҫ������NH3��N2��H2���뿪���������˵��¶�Ӧ�ÿ�����________�森
![]()
�𰸣�
������
������
|
����(1)78��(��4/5) ����(2)H2O ����(3)�Ȱ�ԭ����ͨ��NaOH��Һ(��ʯ��ˮ�ȼ�Һ)����ͨ��Ũ����(���ʯ�ҵȸ����) ����(4)N2��3H2 ����(5)?33.35 |

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ



 CO+H2��CO+H2O
CO+H2��CO+H2O

 CO+H2��CO+H2O
CO+H2��CO+H2O CO2+H2������������ѧ����ʽ�б��������Ե�������______���ѧʽ����
CO2+H2������������ѧ����ʽ�б��������Ե�������______���ѧʽ����