��Ŀ����
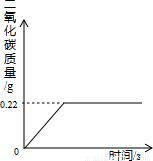
�ڡ���ʽ�����Ƽ���Ĺ��������У����һ�����ü���̼�����Ƶķ�������ȡ���ij�����������ƵõIJ�ƷNa2CO3�л�������NaHCO3��Ϊ�˲ⶨ��Ʒ��Na2CO3������������ȡ100g��������(2NaHCO3 Na2CO3+CO2��+H2O, Na2CO3���Ȳ��ֽ⣩����Ӧ���������ɶ�����̼����������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ��
Na2CO3+CO2��+H2O, Na2CO3���Ȳ��ֽ⣩����Ӧ���������ɶ�����̼����������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ��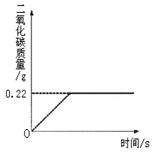
�����������⣺
��1����Ӧ���������ɶ�����̼������Ϊ g
��2��100g�������NaHCO3��������
��1��0.22 ��2��0.84
����

��ϰ��ϵ�д�
 ��ѧ��������������Ͼ���ѧ������ϵ�д�
��ѧ��������������Ͼ���ѧ������ϵ�д� �ϴ�̸�������������νӽ̳��Ͼ���ѧ������ϵ�д�
�ϴ�̸�������������νӽ̳��Ͼ���ѧ������ϵ�д�
�����Ŀ
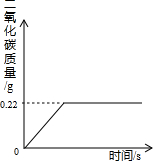
 �ڡ���ʽ�����Ƽ���Ĺ��������У����һ�����ü���̼�����Ƶķ�������ȡ���ij�����������ƵõIJ�ƷNa2CO3�л�������NaHCO3��Ϊ�˲ⶨ��Ʒ��Na2CO3������������ȡ100g�������ȣ�2NaHCO3
�ڡ���ʽ�����Ƽ���Ĺ��������У����һ�����ü���̼�����Ƶķ�������ȡ���ij�����������ƵõIJ�ƷNa2CO3�л�������NaHCO3��Ϊ�˲ⶨ��Ʒ��Na2CO3������������ȡ100g�������ȣ�2NaHCO3 Na2CO3+H2O+CO2����Na2CO3���Ȳ��ֽ⣩����Ӧ���������ɶ�����̼����������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ��
Na2CO3+H2O+CO2����Na2CO3���Ȳ��ֽ⣩����Ӧ���������ɶ�����̼����������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ��
�����������⣺
��1��100g�������NaHCO3�����������Ƕ��٣�
��2����Ӧ������̼���Ƶ������Ƕ��٣�
��3��С��Ҫ����20��ʱNa2CO3�ı�����Һ���������ϣ��õ��±���
| �¶��ܽ���� | 10�� | 20�� | 30�� | 40�� |
| Na2CO3 | 12.5g | 21.5g | 39.7g | 49.0g |
| NaHCO3 | 8.1g | 9.6g | 11.1g | 12.7g |
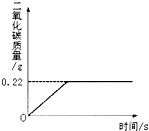
 �ڡ���ʽ�����Ƽ���Ĺ��������У����һ�����ü���̼�����Ƶķ�������ȡ���ij�����������ƵõIJ�ƷNa2CO3�л�������NaHCO3��Ϊ�˲ⶨ��Ʒ��Na2CO3������������ȡ100g�������ȣ�2NaHCO3
�ڡ���ʽ�����Ƽ���Ĺ��������У����һ�����ü���̼�����Ƶķ�������ȡ���ij�����������ƵõIJ�ƷNa2CO3�л�������NaHCO3��Ϊ�˲ⶨ��Ʒ��Na2CO3������������ȡ100g�������ȣ�2NaHCO3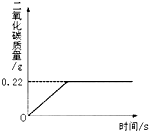 ��2008?��ƽ��һģ���ڡ���ʽ�����Ƽ���Ĺ��������У����һ�����ü���̼�����Ƶķ�������ȡ���ij�����������ƵõIJ�ƷNa2CO3�л�������NaHCO3��Ϊ�˲ⶨ��Ʒ��Na2CO3������������ȡ100g�������ȣ�2NaHCO3
��2008?��ƽ��һģ���ڡ���ʽ�����Ƽ���Ĺ��������У����һ�����ü���̼�����Ƶķ�������ȡ���ij�����������ƵõIJ�ƷNa2CO3�л�������NaHCO3��Ϊ�˲ⶨ��Ʒ��Na2CO3������������ȡ100g�������ȣ�2NaHCO3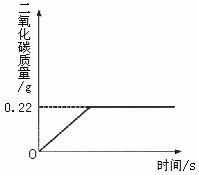
 Na2CO3+H2O+CO2����Na2CO3���Ȳ��ֽ⣩����Ӧ���������ɶ�����̼����������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ��
Na2CO3+H2O+CO2����Na2CO3���Ȳ��ֽ⣩����Ӧ���������ɶ�����̼����������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ��