��Ŀ����
������ԭ����ͭʵ��õ��Ĺ����ĩΪ����ͭ��ͭ�Ļ�����ѧ��ȤС���ͬѧ�Ը÷�ĩ�е���ͭ�ĺ������вⶨ������ͬѧ��ȡ10�˷�ĩ��Ʒ�����ձ��У�Ȼ����벻ͬ������ͬŨ�ȵ�ϡ���ᣬ��ַ�Ӧ���ձ���ʣ�������г�����ʵ�����ݼ�¼���£�| ����� | ��Ʒ�������ˣ� | ����ϡ�����������ˣ� | ʣ������������ˣ� |
| 1 | 10 | 20 | 8.4 |
| 2 | 10 | 30 | 8 |
| 3 | 10 | 40 | 8 |
��2����Ʒ�е���ͭ����������Ϊ______��
��3�������ϡ�����������������Ƕ��٣���д��������̣�
���𰸡��������������йػ�ѧ����ʽ�ļ����⣬�������ڱ����ͼ����⣮������Ŀ��ʽ��������Ҫ�������ࣺһ�Ǹ��������غ㶨�ɣ�������ֵ�����壻���Ǽ��ٵ����������Dzμӷ�Ӧ�Ĺ����������������ڵڶ������ͣ����������֪����һ��������ȫ��Ӧ���ڶ�������������ʣ�࣬����ͭȫ����Ӧ��ֻҪ���������������ݣ�������ϵ������ͻ�ӭ�ж��⣮
����⣺��1������Ŀ��������Ϣ��֪����һ��������ȫ��Ӧ���ڶ�������������ʣ�࣬����ͭȫ����Ӧ��ʣ��Ĺ���ֻ��ͭ���ʴ�Ϊ��ͭ
��2������Ŀ��������Ϣ��֪ͭ��������8g��ͭ����������=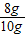 ×100%=80%���ʴ�Ϊ��80%
×100%=80%���ʴ�Ϊ��80%
��3���ɱ�����Ϣ��֪����һ��������ȫ��Ӧ���ڶ�������������ʣ�࣬�����Ҫ�õ�һ�ε����ݣ�
�����������Ϊx
CuO+H2SO4=CuSO4+H2O
80 98
10g-8.4g x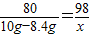 x=1.96g
x=1.96g
�������������=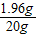 ×100%=9.8% ��ϡ������������������9.8%��
×100%=9.8% ��ϡ������������������9.8%��
�ʴ�Ϊ��9.8%
���������������йػ�ѧ����ʽ�ļ����⣬���ڱ����ͼ����⣮��������п������⣬���Ͷ�������Ҫ�б����⡢ͼ���⡢��ǩ�⡢ͼ��ͱ�������ϵ���ۺ����Լ�������������ȣ��Ӳ�ͬ�ķ��濼����ѧ����ʶͼ���������������ͼ���������ͬѧ��Ҫ������գ�
����⣺��1������Ŀ��������Ϣ��֪����һ��������ȫ��Ӧ���ڶ�������������ʣ�࣬����ͭȫ����Ӧ��ʣ��Ĺ���ֻ��ͭ���ʴ�Ϊ��ͭ
��2������Ŀ��������Ϣ��֪ͭ��������8g��ͭ����������=
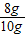 ×100%=80%���ʴ�Ϊ��80%
×100%=80%���ʴ�Ϊ��80%��3���ɱ�����Ϣ��֪����һ��������ȫ��Ӧ���ڶ�������������ʣ�࣬�����Ҫ�õ�һ�ε����ݣ�
�����������Ϊx
CuO+H2SO4=CuSO4+H2O
80 98
10g-8.4g x
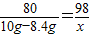 x=1.96g
x=1.96g�������������=
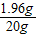 ×100%=9.8% ��ϡ������������������9.8%��
×100%=9.8% ��ϡ������������������9.8%���ʴ�Ϊ��9.8%
���������������йػ�ѧ����ʽ�ļ����⣬���ڱ����ͼ����⣮��������п������⣬���Ͷ�������Ҫ�б����⡢ͼ���⡢��ǩ�⡢ͼ��ͱ�������ϵ���ۺ����Լ�������������ȣ��Ӳ�ͬ�ķ��濼����ѧ����ʶͼ���������������ͼ���������ͬѧ��Ҫ������գ�

��ϰ��ϵ�д�
�����Ŀ

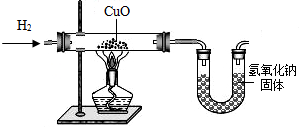
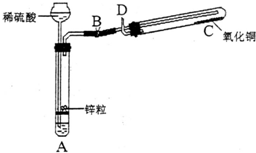 ijѧ������ͼ��ʾװ���Ʊ������������Ƶõ�������ԭ����ͭ��װ���б�Ҫ������̨�����С������豸����ͼ�о�����ȥ��������д���пհף�
ijѧ������ͼ��ʾװ���Ʊ������������Ƶõ�������ԭ����ͭ��װ���б�Ҫ������̨�����С������豸����ͼ�о�����ȥ��������д���пհף�