��Ŀ����
ij��ȤС����д����ᴿ������NaCl��Һ����ش��������⣺

��1�������٢ڢ۶�Ҫ�õ���һ�ֲ���������______��
��2������������Ҫ��Բ����ֽ�۵�����������ͼʾ�в��ó��ֵ�������______������ţ���
��3���������У�����������______ʱ��ֹͣ���ȣ�
��4��ʵ������������õľ��Σ������㾫�εIJ��ʣ����ֲ��ʽϵͣ������ԭ����______������ţ���
A������ʱ��ֽ�����𡡡���������������������B������ʱ�й��彦��
C���ܽ⺬����ɳ�Ĵ�ʳ��ʱ�������ˮ�����㡡D�����������þ��κܳ�ʪ
��5��������ʵ������NaCl��������50g 10%NaCl��Һ������ʱ��ȷ�IJ�����______������ţ���
A���ȷ����룬���ҩƷ������������������ B���ȷ�ҩƷ���������
��6���������õ���Һ������������10%������ܵ�ԭ����______������ţ���
A������Ͳ��ȡˮʱ���Ӷ������������������� B��������Һʱ�ձ�����������ˮ��ϴ
C���Ȼ���δ��ȫ�ܽ�ͽ���Һת�����Լ�ƿ�С�D������ȱ��
��2��A��B��C ����ֽ����ȷ���۵�������D�е���ֽ���ӿڴ����ַ�϶��Һ���ֱ�ӵĽ���������ձ����ʲ�Ӧ�ó��֣�
��3��������������ʱ�����������г��ֽ϶�Ĺ���ʱ��Ӧֹͣ���ȣ�
��4��A������ʱ��ֽ�����𣬻������ɳ������Һ�У��������ε�����������ֵƫ�������Ƶ��ʽϸߣ����������⣻
B������ʱ�й��彦���������������٣������Ƶ��ʽϵͣ��������⣻
C���ܽ⺬����ɳ�Ĵ�ʳ��ʱ�������ˮ�����㣬�ᵼ�������Һ��ʳ�ε������٣������������٣������Ƶ��ʽϵͣ��������⣻
D�����������þ��κܳ�ʪ�����в���ˮ�֣���ɾ����������ӣ������Ƶ��ʽϸߣ����������⣮
��ѡBC��
��5����������ƽ����һ��������ҩƷʱ��Ӧ�ȷ����룬���ҩƷ��ֱ����ƽƽ�⣮
��6��A������Ͳ��ȡˮʱ���Ӷ���������ƫ��ʵ����ȡ����ˮƫС�����ʲ��䣬�ܼ�ƫС��������������ƫ��
B��������Һ���ձ�����������ˮ��ϴ��ʹ��ȡˮ�������ӣ����ʲ��䣬�ܼ�ƫ����������������ƫС��
C���Ȼ���δ��ȫ�ܽ�ͽ���Һת�����Լ�ƿ�У��ᵼ�����������Һ�����ʵ�����ƫС���ܼ����䣬�����õ���Һ��������ƫС��
D������ȱ�𣬻ᵼ������ȡ�����ʵ�����ƫС���ܼ����䣬����������������ƫС��
�ʴ�Ϊ����1������������2��D����3�����ֽ϶�Ĺ��壻��4��BC����5��A����6��A��
��������1�����ݴ����ᴿ�IJ�����з������
��2������ͼʾ�е���ֽ���۵��������з����жϣ�
��3����������������ע��������з������
��4�����ݾ����Ƶ���=
 ��100%��������
��100%���з��������5��������������ƽ����һ������ҩƷ�ķ������з����жϣ�
��6�����������õ���Һ������������10%��ԭ������ǣ��������ʳƶ��ˡ���ȡˮʱ�����ˣ���ˮʱ���Ӷ������ȣ�
�����������ѶȲ��Ǻܴ��ۺ��Խ�ǿ�����մ����ᴿʵ����й�ע�����Ӱ����������������С�����ص�����ȷ�����Ĺؼ���

 ��У����ϵ�д�
��У����ϵ�д���ȤС��ι�ij�Ƽ���������Ϣ���������������о���
���������ϡ�
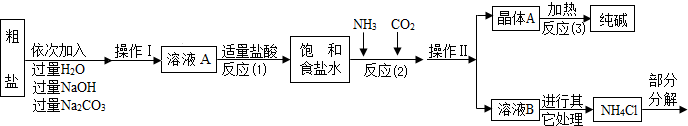
�ٴ����к����������������ʣ�MgCl2��CaCl2�������������ʣ�
�ڷ�Ӧԭ����NaCl�����ͣ�+NH3+CO2+H2O=NaHCO3��+NH4Cl����������ľ���A��ּ��ȣ����Ƶô��
��NH4Cl NH3��+HCl����
NH3��+HCl����
����ˮ����ͭ��ˮ����
�ݲ���������������ͼ��ʾ��
���������ۡ�
��1����д����������������Һ��������Ӧ�Ļ�ѧ����ʽ______��
�ڲ����������Ϊ______��
�۷�Ӧ��1���м����������������______��
�ܷ�Ӧ��2����Ϊ��߲��ʣ����������˳����______������ĸ����
A����ͨ�������̼��ͨ�������� B����ͨ�백����ͨ������̼
��2���������������в���ѭ��ʹ�õ���______������ĸ����
A��CO2��������B��NH3��������C��HCl�������� D��NaOH
�����̽��һ��
��3���پ���A���ȷֽ�Ļ�ѧ����ʽΪ______��
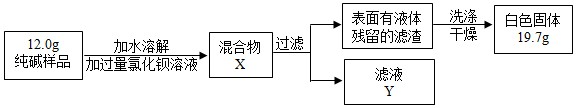
�����ʵ����鴿����Ʒ���Ƿ���о���A������±���
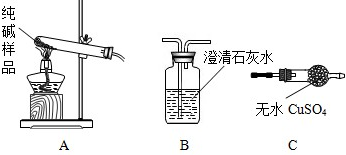
| ѡ���װ�� | ʵ������ | ʵ����� |
| ______ | ______ | ��Ʒ��������A |
��4��ȡ������Ʒ��ˮ�ܽ⣬�����м������ϡHNO3���ٵμ�AgNO3��Һ���а�ɫ���������������ķ���ʽΪ______��ȷ��������Ʒ��������NaCl��
�����̽������
��5��ͬѧ��Ϊ�˲ⶨ�ô�����Ʒ�Ĵ��ȣ����������ʵ�飺

���жϼ���BaCl2��Һ�Ƿ�����ĺ��ʷ�����______���۲������жϣ�
A�����û����X�����ϲ���Һ���ٵ�BaCl2��Һ
B������ҺY�еμ�BaCl2��Һ
���ж������Ƿ�ϴ�Ӹɾ����ɲ�ȡ��ϴ��Һ�еμ�______���۲������жϣ�
A��BaCl2��Һ������ B��ϡH2SO4������C��Na2CO3��Һ����D��ϡHCl
�۸���ʵ�����ݣ�������Ʒ��̼���Ƶ���������Ϊ______�� ��д��������̣�
��ȤС��ι�ij�Ƽ���������Ϣ���������������о���
���������ϡ�
�ٴ����к����������������ʣ�MgCl2��CaCl2�������������ʣ�
�ڷ�Ӧԭ����NaCl�����ͣ�+NH3+CO2+H2O=NaHCO3��+NH4Cl����������ľ���A��ּ��ȣ����Ƶô��
��NH4Cl NH3��+HCl����
NH3��+HCl����
����ˮ����ͭ��ˮ����
�ݲ���������������ͼ��ʾ��

���������ۡ�
��1����д����������������Һ��������Ӧ�Ļ�ѧ����ʽ��������
�ڲ����������Ϊ����
�۷�Ӧ��1���м��������������������
�ܷ�Ӧ��2����Ϊ��߲��ʣ����������˳��������������ĸ����
A����ͨ�������̼��ͨ���� B����ͨ�백����ͨ������̼
��2���������������в���ѭ��ʹ�õ�������������ĸ����
A��CO2 B��NH3 C��HCl D��NaOH
�����̽��һ��
��3���پ���A���ȷֽ�Ļ�ѧ����ʽΪ��������
�����ʵ����鴿����Ʒ���Ƿ���о���A������±���

| ѡ���װ�� | ʵ������ | ʵ����� |
| ���� | �� | ��Ʒ��������A |
�����̽������
��4��ȡ������Ʒ��ˮ�ܽ⣬�����м������ϡHNO3���ٵμ�AgNO3��Һ���а�ɫ���������������ķ���ʽΪ����ȷ��������Ʒ��������NaCl��
�����̽������
��5��ͬѧ��Ϊ�˲ⶨ�ô�����Ʒ�Ĵ��ȣ����������ʵ�飺

���жϼ���BaCl2��Һ�Ƿ�����ĺ��ʷ������������۲������жϣ�
A�����û����X�����ϲ���Һ���ٵ�BaCl2��Һ
B������ҺY�еμ�BaCl2��Һ
���ж������Ƿ�ϴ�Ӹɾ����ɲ�ȡ��ϴ��Һ�еμ��������۲������жϣ�
A��BaCl2��Һ B��ϡH2SO4 C��Na2CO3��Һ D��ϡHCl
�۸���ʵ�����ݣ�������Ʒ��̼���Ƶ���������Ϊ�� ��д��������̣�
���������ϡ�
�ٴ����к����������������ʣ�MgCl2��CaCl2�������������ʣ�
�ڷ�Ӧԭ����NaCl�����ͣ�+NH3+CO2+H2O=NaHCO3��+NH4Cl����������ľ���A��ּ��ȣ����Ƶô��
��NH4Cl
 NH3��+HCl����
NH3��+HCl��������ˮ����ͭ��ˮ����
�ݲ���������������ͼ��ʾ��

���������ۡ�
��1����д����������������Һ��������Ӧ�Ļ�ѧ����ʽ ��
�ڲ����������Ϊ ��
�۷�Ӧ��1���м���������������� ��
�ܷ�Ӧ��2����Ϊ��߲��ʣ����������˳���� ������ĸ����
A����ͨ�������̼��ͨ���� B����ͨ�백����ͨ������̼
��2���������������в���ѭ��ʹ�õ��� ������ĸ����
A��CO2 B��NH3 C��HCl D��NaOH
�����̽��һ��
��3���پ���A���ȷֽ�Ļ�ѧ����ʽΪ ��
�����ʵ����鴿����Ʒ���Ƿ���о���A������±���

| ѡ���װ�� | ʵ������ | ʵ����� |
| ��Ʒ��������A |
��4��ȡ������Ʒ��ˮ�ܽ⣬�����м������ϡHNO3���ٵμ�AgNO3��Һ���а�ɫ���������������ķ���ʽΪ ��ȷ��������Ʒ��������NaCl��
�����̽������
��5��ͬѧ��Ϊ�˲ⶨ�ô�����Ʒ�Ĵ��ȣ����������ʵ�飺

���жϼ���BaCl2��Һ�Ƿ�����ĺ��ʷ����� ���۲������жϣ�
A�����û����X�����ϲ���Һ���ٵ�BaCl2��Һ
B������ҺY�еμ�BaCl2��Һ
���ж������Ƿ�ϴ�Ӹɾ����ɲ�ȡ��ϴ��Һ�еμ� ���۲������жϣ�
A��BaCl2��Һ B��ϡH2SO4 C��Na2CO3��Һ D��ϡHCl
�۸���ʵ�����ݣ�������Ʒ��̼���Ƶ���������Ϊ ��д��������̣�
���������ϡ�
�ٴ����к����������������ʣ�MgCl2��CaCl2�������������ʣ�
�ڷ�Ӧԭ����NaCl�����ͣ�+NH3+CO2+H2O=NaHCO3��+NH4Cl����������ľ���A��ּ��ȣ����Ƶô��
��NH4Cl
 NH3��+HCl����
NH3��+HCl��������ˮ����ͭ��ˮ����
�ݲ���������������ͼ��ʾ��

���������ۡ�
��1����д����������������Һ��������Ӧ�Ļ�ѧ����ʽ ��
�ڲ����������Ϊ ��
�۷�Ӧ��1���м���������������� ��
�ܷ�Ӧ��2����Ϊ��߲��ʣ����������˳���� ������ĸ����
A����ͨ�������̼��ͨ���� B����ͨ�백����ͨ������̼
��2���������������в���ѭ��ʹ�õ��� ������ĸ����
A��CO2 B��NH3 C��HCl D��NaOH
�����̽��һ��
��3���پ���A���ȷֽ�Ļ�ѧ����ʽΪ ��
�����ʵ����鴿����Ʒ���Ƿ���о���A������±���

| ѡ���װ�� | ʵ������ | ʵ����� |
| ��Ʒ��������A |
��4��ȡ������Ʒ��ˮ�ܽ⣬�����м������ϡHNO3���ٵμ�AgNO3��Һ���а�ɫ���������������ķ���ʽΪ ��ȷ��������Ʒ��������NaCl��
�����̽������
��5��ͬѧ��Ϊ�˲ⶨ�ô�����Ʒ�Ĵ��ȣ����������ʵ�飺

���жϼ���BaCl2��Һ�Ƿ�����ĺ��ʷ����� ���۲������жϣ�
A�����û����X�����ϲ���Һ���ٵ�BaCl2��Һ
B������ҺY�еμ�BaCl2��Һ
���ж������Ƿ�ϴ�Ӹɾ����ɲ�ȡ��ϴ��Һ�еμ� ���۲������жϣ�
A��BaCl2��Һ B��ϡH2SO4 C��Na2CO3��Һ D��ϡHCl
�۸���ʵ�����ݣ�������Ʒ��̼���Ƶ���������Ϊ ��д��������̣�