��Ŀ����
��ѧʵ�鿼���У�����������ȡ���ڶ�����̼����ȡ��������50g5%��NaCl��Һ����������ǩ����ѧ����ǩȷ��һ��������п��飮

��1����ͬѧ��ǩ������ʵ���ң����ֱ���ʵ��������Ҫ��������������ҩƷ��
����ͼ������D��������______��F��������______��
�ڼ�ͬѧ�鵽�Ŀ�ǩӦ����______������ĸ���ţ���
A����������ȡ B��������̼����ȡ
����ȡ������ķ�Ӧԭ��Ϊ______���û�ѧ����ʽ��ʾ������ȡһƿ�����壬Ӧѡ�õ�������______������ĸ���ţ���
�ܼ�ͬѧʵ�����Ҫ����ʾ�����£������������������
���װ�á����������ԡ�______���ռ����壮
�������������巢��װ�û�������ȡ�������壬��д������һ�ַ�Ӧ�Ļ�ѧ����ʽ��______��
��2����ͬѧ����ɡ�����50g5%��NaCl��Һ��ʵ������У�ͨ�����㣬���ȡ______g����ȡˮԼ______ml���ܽ�NaClʱ�õ��IJ�������������______��
�⣺��1��������D���������ձ���F�������Ǿƾ��ƣ�
�ڸ��ݸ�����ҩƷϡ����ʹ���ʯ����������ͬѧ�鵽�Ŀ�ǩӦ���ǣ�������̼����ȡ��
����ȡ������̼��ҩƷ��ʯ��ʯ��ϡ���ᣬ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼����Ӧ�ķ���ʽΪ��CaCO3+2HCl=CaCl2+H2O+CO2��������װ���ǹ����Һ���ڳ����µķ�Ӧ����Ҫ�������У�AB����Ϊ������̼������ˮ���ܶȱȿ����ʲ��������ſ������ռ�����Ҫ�������У�CE��
�ܸ÷���ȡ������̼�IJ����ǣ����װ�á����װ�õ������ԡ�װҩƷ���ռ����壻
�ݷ���װ�����ڹ�Һ�����͵�װ�ã���˿�����ʵ�����ù���������Һ�Ͷ���������ȡ���������ж��������Ǵ�������Ӧ�Ļ�ѧ����ʽΪ��2H2O2 2H2O+O2����
2H2O+O2����
��2������50g5%��NaCl��Һ����Ҫ�Ȼ��Ƶ�����=50g��5%=2.5g����Ҫˮ������=50g-2.5g=47.5g���ܽ�NaClʱ�õ��IJ������������Ǽ����ܽ⣻
�ʴ�Ϊ����1�����ձ����ƾ��ƣ���B����CaCO3+2HCl=CaCl2+H2O+CO2����ABCE��������ҩƷ����2H2O2 2H2O+O2����
2H2O+O2����
��2��2.5��47.5�������ܽ⣮
��������1�����������ճ����Ļ�ѧ�������ƺ���;��
�ڸ������������ƺ�ҩƷ����ѡ��
����ȡ������̼��ҩƷ�Ǵ���ʯ��ϡ���ᣬ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼��д����Ӧ�ķ���ʽ�����ݹ����Һ���ڳ����µķ�Ӧѡ����װ�úͶ�����̼���ܶȺ��ܽ���ѡ���ռ���������Ӧ��������
�ܸ���ʵ��IJ��������
�ݸ��ݷ���װ�����ڹ�Һ������װ�ÿ�����ȡ������
ʵ�����ù���������Һ�Ͷ���������ȡ���������������Ǵ�����д����Ӧ�Ļ�ѧ����ʽ��
��2��������Һ�����ʡ��ܼ����������������ļ��㹫ʽ���м��㲢���������������ã�
���������⿼���˳����������ȡװ�á��ռ������ȵ�ѡ���й���Һ�ļ��㣬���ڴ˻����Ͻ�����չ���ؼ�����ȷ����װ�á��ռ�����ѡ������ݣ�
�ڸ��ݸ�����ҩƷϡ����ʹ���ʯ����������ͬѧ�鵽�Ŀ�ǩӦ���ǣ�������̼����ȡ��
����ȡ������̼��ҩƷ��ʯ��ʯ��ϡ���ᣬ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼����Ӧ�ķ���ʽΪ��CaCO3+2HCl=CaCl2+H2O+CO2��������װ���ǹ����Һ���ڳ����µķ�Ӧ����Ҫ�������У�AB����Ϊ������̼������ˮ���ܶȱȿ����ʲ��������ſ������ռ�����Ҫ�������У�CE��
�ܸ÷���ȡ������̼�IJ����ǣ����װ�á����װ�õ������ԡ�װҩƷ���ռ����壻
�ݷ���װ�����ڹ�Һ�����͵�װ�ã���˿�����ʵ�����ù���������Һ�Ͷ���������ȡ���������ж��������Ǵ�������Ӧ�Ļ�ѧ����ʽΪ��2H2O2
 2H2O+O2����
2H2O+O2������2������50g5%��NaCl��Һ����Ҫ�Ȼ��Ƶ�����=50g��5%=2.5g����Ҫˮ������=50g-2.5g=47.5g���ܽ�NaClʱ�õ��IJ������������Ǽ����ܽ⣻
�ʴ�Ϊ����1�����ձ����ƾ��ƣ���B����CaCO3+2HCl=CaCl2+H2O+CO2����ABCE��������ҩƷ����2H2O2
 2H2O+O2����
2H2O+O2������2��2.5��47.5�������ܽ⣮
��������1�����������ճ����Ļ�ѧ�������ƺ���;��
�ڸ������������ƺ�ҩƷ����ѡ��
����ȡ������̼��ҩƷ�Ǵ���ʯ��ϡ���ᣬ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼��д����Ӧ�ķ���ʽ�����ݹ����Һ���ڳ����µķ�Ӧѡ����װ�úͶ�����̼���ܶȺ��ܽ���ѡ���ռ���������Ӧ��������
�ܸ���ʵ��IJ��������
�ݸ��ݷ���װ�����ڹ�Һ������װ�ÿ�����ȡ������
ʵ�����ù���������Һ�Ͷ���������ȡ���������������Ǵ�����д����Ӧ�Ļ�ѧ����ʽ��
��2��������Һ�����ʡ��ܼ����������������ļ��㹫ʽ���м��㲢���������������ã�
���������⿼���˳����������ȡװ�á��ռ������ȵ�ѡ���й���Һ�ļ��㣬���ڴ˻����Ͻ�����չ���ؼ�����ȷ����װ�á��ռ�����ѡ������ݣ�

��ϰ��ϵ�д�
�����Ŀ
 ����Уʵ����ʵʩ��������2011���п���ѧʵ���������ո������Ļ��ʵ�鿼����H2O2�ֽ�������Ϊ�β�����MnO2��������ͬѧ��Ϊ֮չ����̽����
����Уʵ����ʵʩ��������2011���п���ѧʵ���������ո������Ļ��ʵ�鿼����H2O2�ֽ�������Ϊ�β�����MnO2��������ͬѧ��Ϊ֮չ����̽����
I��������������ʵ�������50mL��6%��H2O2��Һ��
[������Һ]
����һƿ�¹��õ�˫��ˮ����ǩ��ͼ��ʾ��
��1��������ƿ˫��ˮ������6%��ҽ��˫��ˮ1500g����Ҫ����˫��ˮ�������Ƕ��ٿˣ�
��2������һƿ5����ǰ��������ͬ����˫��ˮ������⣬�ѻ����ֽ�ų���9.6g������������ʣ��ĸ�ƿ��Һ�����ʵ�����������
II��Ȼ��ͬѧ���ֱȽ��˸������������¶�50mL��6%��H2O2��Һ�ֽⷴӦ�Ĵ����������
[ʵ���¼]��������50mL��6%��H2O2��Һ�ֽⷴӦ�Ĵ��������������22�棩
| ���������� | O2 ƽ������ | ��������ʱ�� | �ɱ� | ��Ӧ��� |
| MnO21.0g | 919mL | 10��03�� | 0.11Ԫ | �ֽ��ȿ���� |
| 10%FeCl3��Һ5�� | 985mL | 4��50�� | 0.02Ԫ | ���Ȳ���O2 |
| 15%CuSO4��Һ5�� | 955mL | 4��57�� | 0.03Ԫ | ���Ȳ���O2 |
[�������]ͬѧ����Ϊ��FeCl3��Һ������Ч����ã���FeCl3��ҺΪ���ܴ��ֽ�H2O2��
[ʵ�����]��֪FeCl3��ˮ�пɽ����Fe3+ ��Cl-��ͬѧ��������²��룺
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�H2O��
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�Fe3+��
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�Cl-��
����Ϊ����ܵ���________ ͬѧ�IJ��룬������________��
ͬѧ�Ƕ����µ��������룬��ʵ�������̽����������ϸ��������գ�
[ʵ�����1]��ʢ��5mL��6% H2O2��Һ���Թ��м���������HCl��Һ�����Ѵ����ǵ�ľ�������Թܣ�
[ʵ������1]����������Ӧ���ۣ�________��
[ʵ�����2]����ʢ��5mL��6%��H2O2��Һ���Թ��м���������________�����ٴΰѴ����ǵ�ľ�������Թܣ�
[ʵ������2]________��
��Ӧ���ۣ�________��
[��չ�о�]
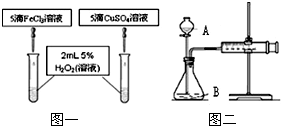
ͬѧ�ǻ��Ƚ���FeCl3��CuSO4�����ζ�H2O2�ֽ�Ĵ�Ч�������Ƿֳɼס������飬�ֱ��������ͼһ��ͼ����ʾ��ʵ�鲢������ʵ�飮������������Һ��Ũ�ȵ�������ͬ��

��1��ָ���������ƣ�A________�� B________
��2��д����FeCl3Ϊ�����ķ�Ӧ�Ļ�ѧ����ʽ________��
��3��ͼһ��ͨ���۲�________�����ԱȽϵó����ۣ���ͬѧ�����ҩƷCuSO4��ΪCuCl2��Ϊ��������������________������ΪҩƷ���������θĽ���________��
��4�����ͼ��װ�������Եķ����ǣ�________������رա�������A�Ļ�������ע������������һ�����룬һ��ʱ����ɿ��������۲쵽�����ֻص���ԭλ����˵��װ��������________������á����á���������ͬѧ����ͼ����ʾʵ���������������________������ţ�A��һ��ʱ�����ռ����������������B���ռ�һ���������������˫��ˮ����������
�о�С�黹����ҺŨ�ȡ��¶ȵ�ʵ������������̽�����±�ѡȡ�˲���ʵ�����ݣ�
�����������H2O2��Һ��ȡ��ͬ���O2�����ʱ��
| Ũ�� ʱ�䣨min�� ���� | 30% H2O2 | 15% H2O2 | 5% H2O2 |
| ����a g MnO2 | 0.2 | 0.8 | 2.0 |
����Уʵ����ʵʩ��������2011���п���ѧʵ���������ո������Ļ��ʵ�鿼����H2O2�ֽ�������Ϊ�β�����MnO2��������ͬѧ��Ϊ֮չ����̽����
I��������������ʵ�������50mL��6%��H2O2��Һ��
[������Һ]
����һƿ�¹��õ�˫��ˮ����ǩ��ͼ��ʾ��
��1��������ƿ˫��ˮ������6%��ҽ��˫��ˮ1500g����Ҫ����˫��ˮ�������Ƕ��ٿˣ�
��2������һƿ5����ǰ��������ͬ����˫��ˮ������⣬�ѻ����ֽ�ų���9.6g������������ʣ��ĸ�ƿ��Һ�����ʵ�����������
II��Ȼ��ͬѧ���ֱȽ��˸������������¶�50mL��6%��H2O2��Һ�ֽⷴӦ�Ĵ����������
[ʵ���¼]��������50mL��6%��H2O2��Һ�ֽⷴӦ�Ĵ��������������22�棩
[ʵ�����]�ɱ������ݼ�ʵ��������Եó���MnO2 ���ֽ�H2O2�����������㲻�㣬�ٿ�ʼ��Ӧ���ʹ��죬������������װ����ѹǿ��Ȼ����ʹ�÷�Һ©����Һ�������裬�ռ�������ˮ���ſ���ʱ�����飬�ͻ��в���O2 ��ʧ�����ռ����һƿO2��Ӧ��������ʱ̫������ ��
[�������]ͬѧ����Ϊ��FeCl3��Һ������Ч����ã���FeCl3��ҺΪ���ܴ��ֽ�H2O2��
[ʵ�����]��֪FeCl3��ˮ�пɽ����Fe3+ ��Cl-��ͬѧ��������²��룺
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�H2O��
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�Fe3+��
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�Cl-��
����Ϊ����ܵ��� ͬѧ�IJ��룬������ ��
ͬѧ�Ƕ����µ��������룬��ʵ�������̽����������ϸ��������գ�
[ʵ�����1]��ʢ��5mL��6% H2O2��Һ���Թ��м���������HCl��Һ�����Ѵ����ǵ�ľ�������Թܣ�
[ʵ������1]����������Ӧ���ۣ� ��
[ʵ�����2]����ʢ��5mL��6%��H2O2��Һ���Թ��м��������� �����ٴΰѴ����ǵ�ľ�������Թܣ�
[ʵ������2] ��
��Ӧ���ۣ� ��
[��չ�о�]
ͬѧ�ǻ��Ƚ���FeCl3��CuSO4�����ζ�H2O2�ֽ�Ĵ�Ч�������Ƿֳɼס������飬�ֱ��������ͼһ��ͼ����ʾ��ʵ�鲢������ʵ�飮������������Һ��Ũ�ȵ�������ͬ��

��1��ָ���������ƣ�A B
��2��д����FeCl3Ϊ�����ķ�Ӧ�Ļ�ѧ����ʽ ��
��3��ͼһ��ͨ���۲� �����ԱȽϵó����ۣ���ͬѧ�����ҩƷCuSO4��ΪCuCl2��Ϊ�������������� ������ΪҩƷ���������θĽ��� ��
��4�����ͼ��װ�������Եķ����ǣ� ������رա�������A�Ļ�������ע������������һ�����룬һ��ʱ����ɿ��������۲쵽�����ֻص���ԭλ����˵��װ�������� ������á����á���������ͬѧ����ͼ����ʾʵ��������������� ������ţ�A��һ��ʱ�����ռ������������ B���ռ�һ���������������˫��ˮ����������
�о�С�黹����ҺŨ�ȡ��¶ȵ�ʵ������������̽�����±�ѡȡ�˲���ʵ�����ݣ�
�����������H2O2��Һ��ȡ��ͬ���O2�����ʱ��
[��������]���ϱ��л��ܵó��Ľ����ǣ� ��

I��������������ʵ�������50mL��6%��H2O2��Һ��
[������Һ]
����һƿ�¹��õ�˫��ˮ����ǩ��ͼ��ʾ��
��1��������ƿ˫��ˮ������6%��ҽ��˫��ˮ1500g����Ҫ����˫��ˮ�������Ƕ��ٿˣ�
��2������һƿ5����ǰ��������ͬ����˫��ˮ������⣬�ѻ����ֽ�ų���9.6g������������ʣ��ĸ�ƿ��Һ�����ʵ�����������
II��Ȼ��ͬѧ���ֱȽ��˸������������¶�50mL��6%��H2O2��Һ�ֽⷴӦ�Ĵ����������
[ʵ���¼]��������50mL��6%��H2O2��Һ�ֽⷴӦ�Ĵ��������������22�棩
| ���������� | O2 ƽ������ | ��������ʱ�� | �ɱ� | ��Ӧ��� |
| MnO21.0g | 919mL | 10��03�� | 0.11Ԫ | �ֽ��ȿ���� |
| 10%FeCl3��Һ5�� | 985mL | 4��50�� | 0.02Ԫ | ���Ȳ���O2 |
| 15%CuSO4��Һ5�� | 955mL | 4��57�� | 0.03Ԫ | ���Ȳ���O2 |
[�������]ͬѧ����Ϊ��FeCl3��Һ������Ч����ã���FeCl3��ҺΪ���ܴ��ֽ�H2O2��
[ʵ�����]��֪FeCl3��ˮ�пɽ����Fe3+ ��Cl-��ͬѧ��������²��룺
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�H2O��
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�Fe3+��
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�Cl-��
����Ϊ����ܵ��� ͬѧ�IJ��룬������ ��
ͬѧ�Ƕ����µ��������룬��ʵ�������̽����������ϸ��������գ�
[ʵ�����1]��ʢ��5mL��6% H2O2��Һ���Թ��м���������HCl��Һ�����Ѵ����ǵ�ľ�������Թܣ�
[ʵ������1]����������Ӧ���ۣ� ��
[ʵ�����2]����ʢ��5mL��6%��H2O2��Һ���Թ��м��������� �����ٴΰѴ����ǵ�ľ�������Թܣ�
[ʵ������2] ��
��Ӧ���ۣ� ��
[��չ�о�]
ͬѧ�ǻ��Ƚ���FeCl3��CuSO4�����ζ�H2O2�ֽ�Ĵ�Ч�������Ƿֳɼס������飬�ֱ��������ͼһ��ͼ����ʾ��ʵ�鲢������ʵ�飮������������Һ��Ũ�ȵ�������ͬ��

��1��ָ���������ƣ�A B
��2��д����FeCl3Ϊ�����ķ�Ӧ�Ļ�ѧ����ʽ ��
��3��ͼһ��ͨ���۲� �����ԱȽϵó����ۣ���ͬѧ�����ҩƷCuSO4��ΪCuCl2��Ϊ�������������� ������ΪҩƷ���������θĽ��� ��
��4�����ͼ��װ�������Եķ����ǣ� ������رա�������A�Ļ�������ע������������һ�����룬һ��ʱ����ɿ��������۲쵽�����ֻص���ԭλ����˵��װ�������� ������á����á���������ͬѧ����ͼ����ʾʵ��������������� ������ţ�A��һ��ʱ�����ռ������������ B���ռ�һ���������������˫��ˮ����������
�о�С�黹����ҺŨ�ȡ��¶ȵ�ʵ������������̽�����±�ѡȡ�˲���ʵ�����ݣ�
�����������H2O2��Һ��ȡ��ͬ���O2�����ʱ��
| Ũ�� ʱ�䣨min�� ���� | 30% H2O2 | 15% H2O2 | 5% H2O2 |
| ����a g MnO2 | 0.2 | 0.8 | 2.0 |

 ����Уʵ����ʵʩ��������2011���п���ѧʵ���������ո������Ļ��ʵ�鿼����H2O2�ֽ�������Ϊ�β�����MnO2��������ͬѧ��Ϊ֮չ����̽����
����Уʵ����ʵʩ��������2011���п���ѧʵ���������ո������Ļ��ʵ�鿼����H2O2�ֽ�������Ϊ�β�����MnO2��������ͬѧ��Ϊ֮չ����̽����