��Ŀ����
���һ����ֹܾ涨��05��1��1������ʵ�������뻷����ܷ�Χ�����꼶ij��ͬѧ�����÷�̪�������������������Һʵ���Һ���е��༶��Һ���У���Һ����ɫ������С��ͬѧ��Ϊ������������������ǡ����ȫ�к͡��Ľ��ۡ�
������Ϊ���ó��Ľ����Ƿ���ȷ��������������������������������������������
��������Һֱ������������ˮ�ܵ��������ʲôΣ��������������������������д����Ӧ����ʽ����������������������������������������������������
����д�����кͷ�Ӧ�Ļ�ѧ����ʽ��������������������������������������������
�������ʵ�飬̽��������Һ���е���Һ�Ƿ�ǡ����ȫ�кͣ���д�±���
| ʵ�鷽�� | ���ܹ۲쵽������ | �ᡡ���� |
| | | |
��1������ȷ����̪��������Һ��ҲΪ��ɫ����2��������ķ�Һֱ������������ˮ�ܵ�����Թܵ���ɸ�ʴ����Ӧ�Ļ�ѧ����ʽΪ��Fe+2HCl=FeCl2+H2����3��NaOH+HCl=NaCl+H2O����4�� ʵ�鷽��
���ձ���ȡ������Ӧ�����Һ��һ֧�Թ��У������Թ��еμ�̼������Һ�����ܹ۲쵽������
1�������ݲ���
2�������ݲ����ᡡ����
����ǡ����ȫ��Ӧ
2��ǡ����ȫ��Ӧ
����

���������Ǵ�����Ⱦ��֮һ����һΪ�ҹ����������������ж�ÿ�ο��������ⶨ�е�S02���Ũ����ֵ��

| S02���Ũ����ֵ����λmg?m-3�� | ||
| һ���� | ������ | ������ |
| 0.15 | 0.50 | 0.70 |
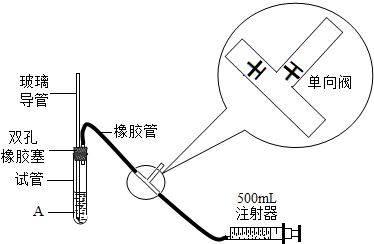
��1�����װ�õ�������ʱ�������Թ���װ��������ˮ����֤�������ܵ��¶˽�û��ˮ�У���Ȼ��������������ע�����Ļ���ʱ�����ῴ��________ ����дʵ��������֤����װ�õ����������ã�
��2����֪�����������ˮ�ķ�ӦΪ��SO2+I2+2H2O�TH2SO4+2HI�����Թ��м���1.0mL������������Ϊ1.27��10-4�ĵ�ˮ�� ��ʱ��Һ���ܶ���ˮ���ܶȽ��� ����������������ˮϡ�ͺ��ټ���2��3����ɫ������Һ�������ⵥ�ʱ���ɫ�������Ƴ���ҺA���ⶨָ���ص�Ŀ�����SO2�ĺ���ʱ������ע�����Ļ�������������A��Һ��________ɫ��Ϊ________ɫʱ��Ӧǡ����ȫ���У�
��3������С��ֳɵ�һС��͵ڶ�С�飬ʹ����ͬ��ʵ��װ�ú���ҺA���ֱ������ѧʵ���Һͽ��ҵĿ����е�S02����������Ӧǡ����ȫ���У���¼�����������£�����ÿ�γ���500mL�����뽫������д����������ʱ������λ��Ч���֣�����I-127��S-32��O-16��
��������ͬ���IJ���ֵ
| �顡�� | ��һС�飨ʵ���ң� | �ڶ�С�飨���ң� |
| �������� | 110 | 150 |
| ������S02�ĺ�������λ��mg?m-3�� |

| S02���Ũ����ֵ | ||
| һ���� | ������ | ������ |
| 0.15 | 0.50 | 0.70 |
��1�����װ�õ�������ʱ�������Թ���װ��������ˮ����֤�������ܵ��¶˽�û��ˮ�У���Ȼ��������������ע�����Ļ���ʱ�����ῴ��______ ����дʵ��������֤����װ�õ����������ã�
��2����֪�����������ˮ�ķ�ӦΪ��SO2+I2+2H2O�TH2SO4+2HI�����Թ��м���1.0mL������������Ϊ1.27×10-4�ĵ�ˮ�� ��ʱ��Һ���ܶ���ˮ���ܶȽ��� ����������������ˮϡ�ͺ��ټ���2��3����ɫ������Һ�������ⵥ�ʱ���ɫ�������Ƴ���ҺA���ⶨָ���ص�Ŀ�����SO2�ĺ���ʱ������ע�����Ļ�������������A��Һ��______ɫ��Ϊ______ɫʱ��Ӧǡ����ȫ���У�
��3������С��ֳɵ�һС��͵ڶ�С�飬ʹ����ͬ��ʵ��װ�ú���ҺA���ֱ������ѧʵ���Һͽ��ҵĿ����е�S02����������Ӧǡ����ȫ���У���¼�����������£�����ÿ�γ���500mL�����뽫������д����������ʱ������λ��Ч���֣�����I-127 S-32 O-16��
���� ��ͬ���IJ���ֵ
| �� �� | ��һС�飨ʵ���ң� | �ڶ�С�飨���ң� |
| �������� | 110 | 150 |
| ������S02��� |