��Ŀ����
��ʵ�����п�����ͼװ������ȡ���壬���dz��õ����巢��װ�ã�ͨ����Һ©����������ƿ�еμ�Һ�壮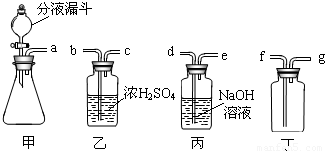
��ش��������⣺
��1��ʵ����ȷ�����巢��װ��ʱӦ���ǵ������� ��
��2����ȡ����ʱ����װ�ü���ƿ�з���������̣��ڷ�Һ©����Ӧ����� ���÷�Ӧ�Ļ�ѧ����ʽΪ ��Һ�巴Ӧ�ﲻ��һ���Լ��뵽��ƿ�У�����ͨ����Һ©����μ��룬���������ŵ��� ��
��3����ȡ���������ʱ��װ�õ���ȷ����˳���ǣ���ܿڵ���ĸ�� ��
��4��װ�ü�Ҳ��������ȡ������̼��ͨ������ �������Ƶõ������Ƿ�Ϊ������̼��д������ʱ��������Ӧ�Ļ�ѧ����ʽ ��
���𰸡���������1��ѡ�����巢��װ��Ҫ���Ƿ�Ӧ���״̬�ͷ�Ӧ������
��2����װ�ü���ȡ����ʱ�����õ�ҩƷ�ǹ���������Һ�Ͷ������̣��ݴ�д����ѧ��Ӧʽ�������ݷ�Һ©�����ص�д���ŵ㣻
��3�����ݳ��õ���������ˮ��װ�ü�ʵ��װ�õ����������
��4������ʯ��ˮ���������������̼���ݷ�Ӧԭ����д����ʽ��
����⣺��1��ȷ�����巢��װ��ʱҪ���Ƿ�Ӧ���״̬�ͷ�Ӧ������
��2����װ�ü���ȡ������Ҫ�õ���ҩƷ�ǹ���������Һ�Ͷ������̣����еĹ���������Һ��Һ�壬Ҫ�ӷ�Һ©���м��룻��ѧ����ʽ�ǣ�2H2O2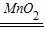 2H2O+O2��������������Һ����μ�����Կ��Ʒ�Ӧ�����ʣ���Ҫʱ�μӣ�����Ҫʱ��ֹͣ�����Ի����Խ�ԼҩƷ��
2H2O+O2��������������Һ����μ�����Կ��Ʒ�Ӧ�����ʣ���Ҫʱ�μӣ�����Ҫʱ��ֹͣ�����Ի����Խ�ԼҩƷ��
��3�����ݳ��õ���������ˮ��װ�ü�ʵ��װ�õ����ӷ�����Ӧ��������ӳ�����ͨ��Ũ���ᣬˮ�ֱ����գ�����������Ӷ̵��ܵ����������ܶȱȿ�����ѡ�������ſ������ռ���Ӧ���������ӳ����ܽ��뼯��ƿ�ײ��������Ӷ̵����ų���
��4�����������̼�����ó���ʯ��ˮ����ͨ���������壬��ʯ��ˮ����ǣ�˵��ͨ��������Ƕ�����̼����Ӧ����ʽ�ǣ�Ca��OH��2+CO2=CaCO3��+H2O��
�ʴ�Ϊ����1����Ӧ���״̬�ͷ�Ӧ������
��2������������Һ��2H2O2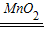 2H2O+O2�����ɿ��Ʒ�Ӧ���ʣ���ԼҩƷ��
2H2O+O2�����ɿ��Ʒ�Ӧ���ʣ���ԼҩƷ��
��3��a��b��c��f��
��4������ʯ��ˮ��Ca��OH��2+CO2=CaCO3��+H2O��
����������װ�õ�ѡ��Ҫ���Ƿ�Ӧ���״̬�ͷ�Ӧ��������Һ©�����Կ��Ʒ�Ӧ�����ʣ�ϴ��װ���е��ܳ����̳������������̼�ó����ʯ��ˮ�ȣ���Щ֪ʶ�ǻ�ѧʵ���еij������⣬ͬѧ��һ��Ҫ���壬���ܽ����������Ľ��
��2����װ�ü���ȡ����ʱ�����õ�ҩƷ�ǹ���������Һ�Ͷ������̣��ݴ�д����ѧ��Ӧʽ�������ݷ�Һ©�����ص�д���ŵ㣻
��3�����ݳ��õ���������ˮ��װ�ü�ʵ��װ�õ����������
��4������ʯ��ˮ���������������̼���ݷ�Ӧԭ����д����ʽ��
����⣺��1��ȷ�����巢��װ��ʱҪ���Ƿ�Ӧ���״̬�ͷ�Ӧ������
��2����װ�ü���ȡ������Ҫ�õ���ҩƷ�ǹ���������Һ�Ͷ������̣����еĹ���������Һ��Һ�壬Ҫ�ӷ�Һ©���м��룻��ѧ����ʽ�ǣ�2H2O2
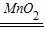 2H2O+O2��������������Һ����μ�����Կ��Ʒ�Ӧ�����ʣ���Ҫʱ�μӣ�����Ҫʱ��ֹͣ�����Ի����Խ�ԼҩƷ��
2H2O+O2��������������Һ����μ�����Կ��Ʒ�Ӧ�����ʣ���Ҫʱ�μӣ�����Ҫʱ��ֹͣ�����Ի����Խ�ԼҩƷ����3�����ݳ��õ���������ˮ��װ�ü�ʵ��װ�õ����ӷ�����Ӧ��������ӳ�����ͨ��Ũ���ᣬˮ�ֱ����գ�����������Ӷ̵��ܵ����������ܶȱȿ�����ѡ�������ſ������ռ���Ӧ���������ӳ����ܽ��뼯��ƿ�ײ��������Ӷ̵����ų���
��4�����������̼�����ó���ʯ��ˮ����ͨ���������壬��ʯ��ˮ����ǣ�˵��ͨ��������Ƕ�����̼����Ӧ����ʽ�ǣ�Ca��OH��2+CO2=CaCO3��+H2O��
�ʴ�Ϊ����1����Ӧ���״̬�ͷ�Ӧ������
��2������������Һ��2H2O2
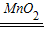 2H2O+O2�����ɿ��Ʒ�Ӧ���ʣ���ԼҩƷ��
2H2O+O2�����ɿ��Ʒ�Ӧ���ʣ���ԼҩƷ����3��a��b��c��f��
��4������ʯ��ˮ��Ca��OH��2+CO2=CaCO3��+H2O��
����������װ�õ�ѡ��Ҫ���Ƿ�Ӧ���״̬�ͷ�Ӧ��������Һ©�����Կ��Ʒ�Ӧ�����ʣ�ϴ��װ���е��ܳ����̳������������̼�ó����ʯ��ˮ�ȣ���Щ֪ʶ�ǻ�ѧʵ���еij������⣬ͬѧ��һ��Ҫ���壬���ܽ����������Ľ��

��ϰ��ϵ�д�
�����Ŀ
 ����ͬѧ����ͼ��ʵ�������Ʊ�������
����ͬѧ����ͼ��ʵ�������Ʊ�������
 ʵ�����п�������ͼװ������ȡ����
ʵ�����п�������ͼװ������ȡ���� 





