��Ŀ����
��8�֣�����װ�ÿ���ȡ�й����塣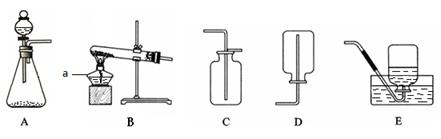
��ش�
��1������a�������� ��
��2��ʵ�����ø��������ȡ�����Ļ�ѧ����ʽΪ ��
Ӧѡ�õķ���װ���� ������ĸ��������Eװ���ռ����������۲쵽
ʱ���ٰѵ��ܿ�����ʢ��ˮ�ļ���ƿ�
��3��ʵ������ȡ������̼�Ļ�ѧ����ʽΪ ��Ӧѡ�õ��ռ�װ���� ������ĸ����
��4����ȡ������̼ʱ��������˽�Ũ�����ᣬ��ʹCO2�л��� �������ƣ�����Ҫ��ȥCO2�еĸ����ʣ�Ӧѡ����ͼװ���е� ������ĸ����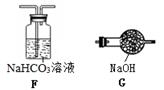
��1���ƾ���
��2��2KMnO4 K2MnO4+ MnO2+O2����B�������������ȽϾ��ȵطų�
K2MnO4+ MnO2+O2����B�������������ȽϾ��ȵطų�
��3��CaCO3+2HCI ="=" CaCl2+H2O+CO2����C
��4���Ȼ��⣻F
����

��ϰ��ϵ�д�
�����Ŀ




 ʵ���ҳ�������װ������ȡ������
ʵ���ҳ�������װ������ȡ������