��Ŀ����
 С��ͬѧΪ�˲ⶨ��������̼��Ƶ���������������������ʵ��̽����
С��ͬѧΪ�˲ⶨ��������̼��Ƶ���������������������ʵ��̽����
��ʵ����̡�С��ͬѧ��װ����ͼװ�ã���������ϴ�������ﲢ����������ƿ�����������ϡ�����ַ�Ӧ�������������ʲ������ᷴӦ��������������������ʾ��
�����ǣ�12.5g��+ϡ���ᣨ40.5g�� ʣ��Ļ���48.6g��
ʣ��Ļ���48.6g��
�����ݴ�����
��1�����������غ㶨�ɣ���Ӧ���ɶ�����̼������Ϊ______g��
��2�����û�ѧ����ʽ���㼦������̼��Ƶ����ʵ����Ƕ��٣�����˵�����̼��Ƶ����������Ƕ��٣���д��������̣�
��ʵ�鷴˼��
�ø�װ����ȡ������̼����Ӧ���нϳ�ʱ���С����ȼ�ŵĻ����ڼ���ƿ�ڣ������Բ�Ϩ�����ʵ��װ���Ϸ���������Ϊԭ����______��
�⣺�����ݴ�������1�����������غ㶨�ɣ���Ӧ���ɶ�����̼������=12.5g+40.5g-48.6g=4.4g
��2����ü�������̼��Ƶ�����Ϊx
CaCO3+2HCl�TCaCl2+CO2��+H2O
100 44
x 4.4g
 =
=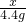 x=10g
x=10g
��������̼��Ƶ����ʵ���= =0.1mol
=0.1mol
�˵�����̼��Ƶ���������= ��100%=80%
��100%=80%
�𣺼�������̼��Ƶ����ʵ�����0.1mol���ü�������̼��Ƶ���������Ϊ80%��
��ʵ�鷴˼����ȼ�ŵĻ����ڼ���ƿ�ڣ������Բ�Ϩ�����ٿ�˵��û���ռ���������̼����ɴ������ԭ��֮һ������ͼ��װ���г���©��δ����Һ�����£����²����Ķ�����̼����ӳ���©���ݳ���
�ʴ�Ϊ������©���¶˹ܿ���Һ�⣬����ӳ���©���ݳ���
�����������ݴ�������1��������������̼��������ᷴӦ�ų�������̼ʹ��Ӧ��ʣ��Ļ����������С�����������غ㶨�ɣ�ʣ��Ļ����������С��������Ӧ�ų�������̼��������
��2������̼��������ᷴӦ�Ļ�ѧ����ʽ���ɷų�������̼�������������Ӧ������̼��Ƶ�����������������ȡ��������Ʒ��������̼����������ɴ˿���ô˵�����̼��Ƶ�����������
��ʵ�鷴˼����ȼ�ŵĻ����ڼ���ƿ�ڣ������Բ�Ϩ�𣬿��ж�û�ռ���������̼����ɴ������ԭ��֮һ��װ��©�����۲�ͼ��װ�ã��ɷ��ֳ���©���IJ���ȷʹ���Dz���װ��©��������ԭ��
�����������ʵ������ݴ���������ԭ��������п��飬���ʱ��Ҫ��ǿ�����ݷ���������ʵ��Ļ������ܣ�
��2����ü�������̼��Ƶ�����Ϊx
CaCO3+2HCl�TCaCl2+CO2��+H2O
100 44
x 4.4g
 =
=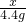 x=10g
x=10g��������̼��Ƶ����ʵ���=
 =0.1mol
=0.1mol�˵�����̼��Ƶ���������=
 ��100%=80%
��100%=80%�𣺼�������̼��Ƶ����ʵ�����0.1mol���ü�������̼��Ƶ���������Ϊ80%��
��ʵ�鷴˼����ȼ�ŵĻ����ڼ���ƿ�ڣ������Բ�Ϩ�����ٿ�˵��û���ռ���������̼����ɴ������ԭ��֮һ������ͼ��װ���г���©��δ����Һ�����£����²����Ķ�����̼����ӳ���©���ݳ���
�ʴ�Ϊ������©���¶˹ܿ���Һ�⣬����ӳ���©���ݳ���
�����������ݴ�������1��������������̼��������ᷴӦ�ų�������̼ʹ��Ӧ��ʣ��Ļ����������С�����������غ㶨�ɣ�ʣ��Ļ����������С��������Ӧ�ų�������̼��������
��2������̼��������ᷴӦ�Ļ�ѧ����ʽ���ɷų�������̼�������������Ӧ������̼��Ƶ�����������������ȡ��������Ʒ��������̼����������ɴ˿���ô˵�����̼��Ƶ�����������
��ʵ�鷴˼����ȼ�ŵĻ����ڼ���ƿ�ڣ������Բ�Ϩ�𣬿��ж�û�ռ���������̼����ɴ������ԭ��֮һ��װ��©�����۲�ͼ��װ�ã��ɷ��ֳ���©���IJ���ȷʹ���Dz���װ��©��������ԭ��
�����������ʵ������ݴ���������ԭ��������п��飬���ʱ��Ҫ��ǿ�����ݷ���������ʵ��Ļ������ܣ�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
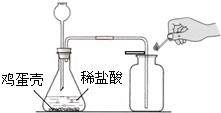 �����ǵ���Ҫ�ɷ���̼��ƣ�С��ͬѧΪ�˲ⶨ��������̼��Ƶ������������ֱ����������ʵ�鲢���м��㣮
�����ǵ���Ҫ�ɷ���̼��ƣ�С��ͬѧΪ�˲ⶨ��������̼��Ƶ������������ֱ����������ʵ�鲢���м��㣮
 ��2010?�ɽ���һģ��С��ͬѧΪ�˲ⶨ��������̼��Ƶ���������������������ʵ��̽����
��2010?�ɽ���һģ��С��ͬѧΪ�˲ⶨ��������̼��Ƶ���������������������ʵ��̽���� ��2012?������һģ�������ǵ���Ҫ�ɷ���̼��ƣ�С��ͬѧΪ�˲ⶨ��������̼��Ƶ��������������������µ�̽��ʵ�飬����������Ļ��
��2012?������һģ�������ǵ���Ҫ�ɷ���̼��ƣ�С��ͬѧΪ�˲ⶨ��������̼��Ƶ��������������������µ�̽��ʵ�飬����������Ļ��
