��Ŀ����
��2010?��̨��һģ���о���ѧϰС��Ϊ�˲ⶨ��ͭ��ͭ��п�Ͻ𣩵���ɣ��õ�����ƽ�ֱ�Ƶ���ƿ����������Ϊ44.1g����ȡ��ͭ��Ʒ20.0g������ƿ�м������Ʒ������ϡ�����ƿ������������ͼ1��ʾ����������ƽ���������ݻ����ͼ2��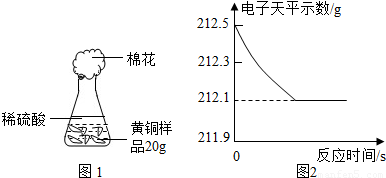
������������ݣ��ش��������⣺
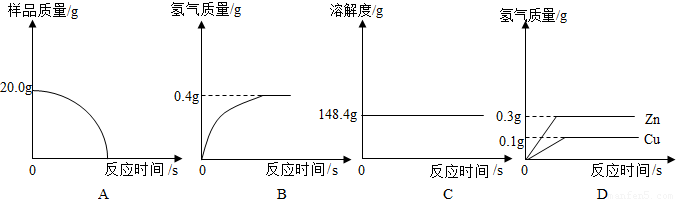
��1���ĸ�ͬѧ�Ӷ�Ƕȴ������ݣ��������ݴ�����ͼ������ȷ����______��
��2���Լ��㣺
����Ʒ��ͭ������������
��ǡ�÷�Ӧʱ������Һ�������������������
���𰸡���������1����Ϊͭ����ϡ���ᷴӦ����ͭ��ֻ��п��ϡ���ᷴӦ����Һ���������ŷ�Ӧ�Ľ��ж��仯��������������������ͼ2�ɼ��������Ȼ�����ѡ����з������ɣ�
��2������п�����ᷴӦ�Ļ�ѧ����ʽ�������������������г�����ʽ���Ϳɼ������Ʒ��п�����������ɵ�����п��������
����Ʒ��ͭ������=��Ʒ����-��Ʒ��п��������Ȼ����ݡ� ×100%�����㼴�ɣ�
×100%�����㼴�ɣ�
��������Һ����������������������ɵ�����п����������Һ����=��ȫ��Ӧ�������ƽʾ��-��ƿ����������-ͭ��������Ȼ�������������������ʽ���㼴�ɣ�
����⣺��1��A����Ϊͭ����ϡ���ᷴӦ����ͭ��ֻ��п��ϡ���ᷴӦ��������ȫ��Ӧ����Ʒ��ʣ�ࣨͭ������A���ʺϣ�
B����ͼ2���ݺ������غ㶨�ɿɵã���ȫ��Ӧʱ��������������Ϊ��212.5g-212.1g=0.4g����B���ϣ�
C����Ϊп��ϡ���ᷴӦ���������������ɣ�������������С�ڲ��뷴Ӧ��п������������Һ���������ŷ�Ӧ�Ľ��ж������ӣ�����Ӧ��ȫʱ���ٱ仯����C���ʺϣ�
D����Ϊͭ����ϡ���ᷴӦ����ͭ��ֻ��п��ϡ���ᷴӦ����D���ʺϣ�
��ѡB��
��2������Ʒ��п������Ϊx�����ɵ�����п������Ϊy��
Zn+H2SO4=ZnSO4+H2��
65 161 2
X y 0.4g
��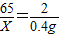 ��
��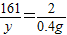 ��
��
��֮�ã�X=13g��y=32.2g��
����Ʒ��ͭ����������Ϊ��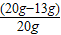 ×100%=35%��
×100%=35%��
��������Һ���������������������Ϊ��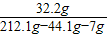 ×100%=20%��
×100%=20%��
�𣺢���Ʒ��ͭ����������Ϊ35%���ڷ�Ӧ��������Һ������п����������Ϊ20%��
������������Ҫ����ѧ�����ۺϷ����������ж����ݴ�����ͼ�������Ҫ�������ʼ䷴Ӧ�������ͼʾ���ݷ���������ʱҪ������֪�����ͻ�ѧ����ʽ�Լ�����������ʽ���У�
��2������п�����ᷴӦ�Ļ�ѧ����ʽ�������������������г�����ʽ���Ϳɼ������Ʒ��п�����������ɵ�����п��������
����Ʒ��ͭ������=��Ʒ����-��Ʒ��п��������Ȼ����ݡ�
 ×100%�����㼴�ɣ�
×100%�����㼴�ɣ���������Һ����������������������ɵ�����п����������Һ����=��ȫ��Ӧ�������ƽʾ��-��ƿ����������-ͭ��������Ȼ�������������������ʽ���㼴�ɣ�
����⣺��1��A����Ϊͭ����ϡ���ᷴӦ����ͭ��ֻ��п��ϡ���ᷴӦ��������ȫ��Ӧ����Ʒ��ʣ�ࣨͭ������A���ʺϣ�
B����ͼ2���ݺ������غ㶨�ɿɵã���ȫ��Ӧʱ��������������Ϊ��212.5g-212.1g=0.4g����B���ϣ�
C����Ϊп��ϡ���ᷴӦ���������������ɣ�������������С�ڲ��뷴Ӧ��п������������Һ���������ŷ�Ӧ�Ľ��ж������ӣ�����Ӧ��ȫʱ���ٱ仯����C���ʺϣ�
D����Ϊͭ����ϡ���ᷴӦ����ͭ��ֻ��п��ϡ���ᷴӦ����D���ʺϣ�
��ѡB��
��2������Ʒ��п������Ϊx�����ɵ�����п������Ϊy��
Zn+H2SO4=ZnSO4+H2��
65 161 2
X y 0.4g
��
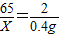 ��
��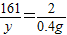 ��
����֮�ã�X=13g��y=32.2g��
����Ʒ��ͭ����������Ϊ��
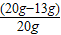 ×100%=35%��
×100%=35%����������Һ���������������������Ϊ��
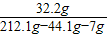 ×100%=20%��
×100%=20%���𣺢���Ʒ��ͭ����������Ϊ35%���ڷ�Ӧ��������Һ������п����������Ϊ20%��
������������Ҫ����ѧ�����ۺϷ����������ж����ݴ�����ͼ�������Ҫ�������ʼ䷴Ӧ�������ͼʾ���ݷ���������ʱҪ������֪�����ͻ�ѧ����ʽ�Լ�����������ʽ���У�

��ϰ��ϵ�д�
 �Ƹ�С״Ԫ�������������ϵ�д�
�Ƹ�С״Ԫ�������������ϵ�д�
�����Ŀ
��2010?��̨��һģ�����й��ڻ�ѧʵ��ġ�Ŀ��һ����һ����һ���ۡ���������ȷ���ǣ� ��
| ʵ����� ��� | ʵ��Ŀ�� | �����Լ� ��������� | ʵ������ | ʵ����� |
| A | ����ij����������Ƿ���CO2���� | ͨ�����ʯ��ˮ�� | ���������� | �������в���CO2 |
| B | ��ȥNaCl�е�����Na2SO4 | �����Ȼ�����Һ | ������ɫ���� | Na2SO4 |
| C | ����Ӳˮ������ˮ | ����ˮ | ����������ĭ | ��Һ��Ϊ����ˮ |
| D | ���ſ������ռ�CO2ʱ���鼯��ƿ��CO2�Ƿ��� | ��ȼ�յ�ľ������ƿ�� | ľ��Ϩ�� | ƿ��CO2�Ѽ��� |
��2010?��̨��һģ�����й��ڻ�ѧʵ��ġ�Ŀ��һ����һ����һ���ۡ���������ȷ���ǣ� ��
| ʵ����� ��� | ʵ��Ŀ�� | �����Լ� ��������� | ʵ������ | ʵ����� |
| A | ����ij����������Ƿ���CO2���� | ͨ�����ʯ��ˮ�� | ���������� | �������в���CO2 |
| B | ��ȥNaCl�е�����Na2SO4 | �����Ȼ�����Һ | ������ɫ���� | Na2SO4 |
| C | ����Ӳˮ������ˮ | ����ˮ | ����������ĭ | ��Һ��Ϊ����ˮ |
| D | ���ſ������ռ�CO2ʱ���鼯��ƿ��CO2�Ƿ��� | ��ȼ�յ�ľ������ƿ�� | ľ��Ϩ�� | ƿ��CO2�Ѽ��� |


 2CuO
2CuO 2CuSO4+2H2O
2CuSO4+2H2O