��Ŀ����
ijͬѧ�ռ������е�һЩ�������ʣ�������������⣺
��1���±��Ǹ�ͬѧ����Щ���ʵķ���������������ڱ�����д���ʵ����ѡ���������������������ᡱ��������Ρ���
��1���±��Ǹ�ͬѧ����Щ���ʵķ���������������ڱ�����д���ʵ����ѡ���������������������ᡱ��������Ρ���
��2��ʳ������________��ѡ����ӡ�����ԭ�ӡ������ӡ�����ͬ�����ɵģ�ͭ����________���ɵġ�
��3��������һ�����ܹ���ˮ�������Ϸ�Ӧ�������ʵĻ�ѧʽΪ________��
��4���������ʯ��¶���ڿ����У����ױ��ʣ���Ӧ�Ļ�ѧ����ʽΪ________________��
��3��������һ�����ܹ���ˮ�������Ϸ�Ӧ�������ʵĻ�ѧʽΪ________��
��4���������ʯ��¶���ڿ����У����ױ��ʣ���Ӧ�Ļ�ѧ����ʽΪ________________��
��1���������ᡢ�Ρ�������
��2�����ӡ�ԭ��
��3��CaO
��4��Ca(OH)2 + CO2===CaCO3�� + H2O
��2�����ӡ�ԭ��
��3��CaO
��4��Ca(OH)2 + CO2===CaCO3�� + H2O

��ϰ��ϵ�д�
�����Ŀ
ijͬѧ�ռ������е�һЩ�������ʣ�������������⣺
��1���±��Ǹ�ͬѧ����Щ���ʵķ���������������ڱ�����д���ʵ����ѡ���������������������ᡱ��������Ρ�
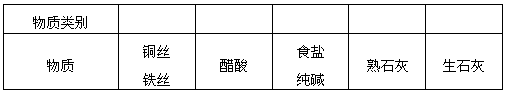
| ������� | ______ | ______ | ______ | ______ | ______ |
| ���� | ͭ˿����˿ | ���� | ʳ�Ρ����� | ��ʯ�� | ��ʯ�� |
��3��������һ�����ܹ���ˮ�������Ϸ�Ӧ�������ʵĻ�ѧʽΪ______��
��4���������ʯ��¶���ڿ����У����ױ��ʣ���Ӧ�Ļ�ѧ����ʽΪ______��
ijͬѧ�ռ������е�һЩ�������ʣ�������������⣺
��1���±��Ǹ�ͬѧ����Щ���ʵķ���������������ڱ�����д���ʵ����ѡ���������������������ᡱ��������Ρ�
��2��ʳ������ ��ѡ����ӡ�����ԭ�ӡ������ӡ�����ͬ�����ɵģ�ͭ���� ���ɵģ�
��3��������һ�����ܹ���ˮ�������Ϸ�Ӧ�������ʵĻ�ѧʽΪ ��
��4���������ʯ��¶���ڿ����У����ױ��ʣ���Ӧ�Ļ�ѧ����ʽΪ ��
��1���±��Ǹ�ͬѧ����Щ���ʵķ���������������ڱ�����д���ʵ����ѡ���������������������ᡱ��������Ρ�
| ������� | |||||
| ���� | ͭ˿����˿ | ���� | ʳ�Ρ����� | ��ʯ�� | ��ʯ�� |
��3��������һ�����ܹ���ˮ�������Ϸ�Ӧ�������ʵĻ�ѧʽΪ ��
��4���������ʯ��¶���ڿ����У����ױ��ʣ���Ӧ�Ļ�ѧ����ʽΪ ��