��Ŀ����
a��ij�¶�ʱ��ij�������ʵ���Һ40g��Ϊ���ȷݣ�һ�ݺ�������ˮ����Һ�ﱥ��ʱ������������һ�룺��һ�ݼ���ԭ���ʣ���Һ�ﱥ��ʱ���������ʵ�����ǡ��Ϊ����Һ�����İ˷�֮һ����
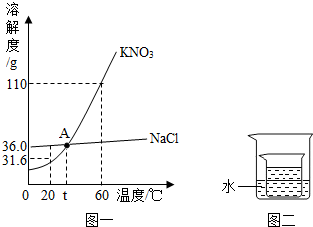
��1�����¶�ʱ�����ʵ��ܽ�ȣ�
��2��ԭ��Һ����������������
b����ʢ��һ��������ͭ��Һ���ձ��е�������������Һ�����������ٲ���ʱ��ȥ����������Һ20g������Ӧ�����Һ���ˣ���4.9g������55��lg��Һ������Һ������12.5gˮ��ǡ���γ�22��ʱ�����Ʊ�����Һ����
��1��ԭ����ͭ��Һ������ͭ����������������
��2��22��ʱ�����Ƶ��ܽ�ȣ�
�⣺
a����1��40g��Һ��Ϊ���ȷݣ�ÿһ����ҺΪ20g��
��һ����Һ�����ͣ�������Ϊ10g��˵��������10gˮ���÷���Һ������Ϊ��10g������Һ��10gˮ��
�ڶ��ݼ����ʣ���20g��1/8����2.5g����Ϊ������Һ��������Һ��ȫһ�£��൱��2.5g�����ܽ���10gˮ��ǡ�ôﵽ���ͣ�����¶������ʵ��ܽ��ΪS�����ԣ�
S��2.5g=100g��10g
S=25g
��2���Ե�һ�ݽ��м��㣮������Ϊ��10g������Һ��10gˮ�Ļ�����10g������Һ��������ΪX������25g��X=125g��10g�� X=2g������ԭ��һ����Һ����������Ϊ��
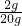 ��100%=10% ��ԭ��Һ��������Ϊ10%��
��100%=10% ��ԭ��Һ��������Ϊ10%��
b���⣺��ԭ����ͭ��Һ�е�����ͭ����Ϊx�����ɵ������Ƶ�����Ϊy��
CuSO4+2NaOH=Cu��OH��2��+Na2SO4
160 98 142
x 4.9g y
160��98=x��4.9g x=8g
98��142=4.9g��y y=7.1g
����ԭ����ͭ��Һ�����ʵ���������Ϊ��
8g/��4.9g+55.1g-20g����100%=20%
��22��ʱ�����Ƶ��ܽ��ΪS��
S��7.1g=100g����55.1g-12.5g-7.1g�� S=20g
��ԭ����ͭ��Һ������ͭ��������������Ϊ20%��22��ʱ�����Ƶ��ܽ��Ϊ20g
�ʴ�Ϊ��
a����1��25g
��2��10%��
b����1��20%
��2��20g��
������������Ŀ��Ϣ�������ܽ�ȵĸ����г���ϵʽ��������ܽ�ȵĴ�С����������������������ʽ���������������������
����������Ĺؼ����ܹ��ҳ�������Һ�����ʺ��ܼ����������Ӷ������ܽ�ȸ����м���ʽ��������������ʡ���Һ����������������������������a����ѣ�ͬѧ��Ҫϸ��˼������ѡһ����ҿ������Լ��ó�����Ŀ���з�����
a����1��40g��Һ��Ϊ���ȷݣ�ÿһ����ҺΪ20g��
��һ����Һ�����ͣ�������Ϊ10g��˵��������10gˮ���÷���Һ������Ϊ��10g������Һ��10gˮ��
�ڶ��ݼ����ʣ���20g��1/8����2.5g����Ϊ������Һ��������Һ��ȫһ�£��൱��2.5g�����ܽ���10gˮ��ǡ�ôﵽ���ͣ�����¶������ʵ��ܽ��ΪS�����ԣ�
S��2.5g=100g��10g
S=25g
��2���Ե�һ�ݽ��м��㣮������Ϊ��10g������Һ��10gˮ�Ļ�����10g������Һ��������ΪX������25g��X=125g��10g�� X=2g������ԭ��һ����Һ����������Ϊ��
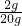 ��100%=10% ��ԭ��Һ��������Ϊ10%��
��100%=10% ��ԭ��Һ��������Ϊ10%��b���⣺��ԭ����ͭ��Һ�е�����ͭ����Ϊx�����ɵ������Ƶ�����Ϊy��
CuSO4+2NaOH=Cu��OH��2��+Na2SO4
160 98 142
x 4.9g y
160��98=x��4.9g x=8g
98��142=4.9g��y y=7.1g
����ԭ����ͭ��Һ�����ʵ���������Ϊ��
8g/��4.9g+55.1g-20g����100%=20%
��22��ʱ�����Ƶ��ܽ��ΪS��
S��7.1g=100g����55.1g-12.5g-7.1g�� S=20g
��ԭ����ͭ��Һ������ͭ��������������Ϊ20%��22��ʱ�����Ƶ��ܽ��Ϊ20g
�ʴ�Ϊ��
a����1��25g
��2��10%��
b����1��20%
��2��20g��
������������Ŀ��Ϣ�������ܽ�ȵĸ����г���ϵʽ��������ܽ�ȵĴ�С����������������������ʽ���������������������
����������Ĺؼ����ܹ��ҳ�������Һ�����ʺ��ܼ����������Ӷ������ܽ�ȸ����м���ʽ��������������ʡ���Һ����������������������������a����ѣ�ͬѧ��Ҫϸ��˼������ѡһ����ҿ������Լ��ó�����Ŀ���з�����

��ϰ��ϵ�д�
�����Ŀ