��Ŀ����
һ��ѧ��ȤС���ij������Ʒ������������������̽������С���������֪�������г����⣬������̼���衢�̡������Ԫ�أ�̼����������������̬���ڣ�һ��������̼��ʯī������һ���ǻ���̼����Fe3C�ȣ����衢�̡��ס��������ʽ��Ϊ���ӣ���Щ����ͨ��������ϡ���ᷢ����ѧ��Ӧ��ʵ��̽������1��ȡһֻ�ձ����Ƶ�������Ϊ72.4g������һ����������Ʒ��м���Ƶ��ձ�����Ʒ��������Ϊ102.4g��
��2����ʢ��������Ʒ���ձ��м���ϡ���ᣬ�ӱ߽��裬ֱ���ձ��в��ٲ������ݣ��ټ�������ϡ������ȷ����������ù���ȥϡ����247.3g��
��3�����ã��������Ƶ��ձ���ʣ�����ʵ�������Ϊ348.7g��
�ش𣺣�����С�����1λС����
��1�������������Ʒ�е�����������������
��2�����йز��ž�ȷ�ⶨ������Ʒ��Fe3C����������Ϊ2%������û�������ᷴӦ��������Fe3C��ʽ���ڣ�����Ʒ��������Ԫ�ص���������Ϊ���٣�
���𰸡���������1���ձ���ǰ����ٵ������������������������������ᷴӦ�Ļ�ѧ����ʽ����������������������������������ٸ���  %���������Ʒ�е�����������������
%���������Ʒ�е�����������������
��2����Ʒ�е���Ԫ�ذ������ʵ�����Fe3C�е��������ݣ�1���Ľ��ۼ���2���������������Ԫ�ص�����������������Ԫ�ص���������Ϊ�� ×100%��⼴�ɣ�
×100%��⼴�ɣ�
����⣺��1������H2��������102.4 g+247.3 g-348.7 g=1.0 g����Ʒ��������102.4g-72.4g=30g
��������Ʒ�к���������Ϊx��
Fe+H2SO4�TFeSO4+H2��
56 2
x 1.0 g
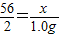
x=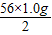 =28.0g
=28.0g
����������������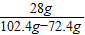 ×100%=93.3%
×100%=93.3%
��2����Ʒ�е���Ԫ�ذ������ʵ�����Fe3C�е�������Ԫ�ص�������=28.0g+30g×2%×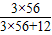 =28.56g��
=28.56g��
��������Ԫ�ص���������Ϊ�� =
=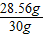 ×100%=95.2%
×100%=95.2%
�𣺣�1����������Ʒ�е�����������������93.3%
��2����Ʒ��������Ԫ�ص���������Ϊ95.2%
�����������Ƕ�������ij��Ԫ�ػ������������Ŀ��飬����Ĺؼ��ǶԷ�Ӧǰ�������仯��Ӧ�ã�����һ�����������ۺ��⣮
 %���������Ʒ�е�����������������
%���������Ʒ�е�������������������2����Ʒ�е���Ԫ�ذ������ʵ�����Fe3C�е��������ݣ�1���Ľ��ۼ���2���������������Ԫ�ص�����������������Ԫ�ص���������Ϊ��
 ×100%��⼴�ɣ�
×100%��⼴�ɣ�����⣺��1������H2��������102.4 g+247.3 g-348.7 g=1.0 g����Ʒ��������102.4g-72.4g=30g
��������Ʒ�к���������Ϊx��
Fe+H2SO4�TFeSO4+H2��
56 2
x 1.0 g
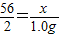
x=
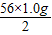 =28.0g
=28.0g����������������
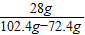 ×100%=93.3%
×100%=93.3%��2����Ʒ�е���Ԫ�ذ������ʵ�����Fe3C�е�������Ԫ�ص�������=28.0g+30g×2%×
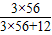 =28.56g��
=28.56g����������Ԫ�ص���������Ϊ��
 =
=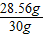 ×100%=95.2%
×100%=95.2%�𣺣�1����������Ʒ�е�����������������93.3%
��2����Ʒ��������Ԫ�ص���������Ϊ95.2%
�����������Ƕ�������ij��Ԫ�ػ������������Ŀ��飬����Ĺؼ��ǶԷ�Ӧǰ�������仯��Ӧ�ã�����һ�����������ۺ��⣮

��ϰ��ϵ�д�
�����Ŀ
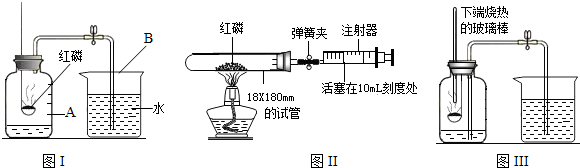
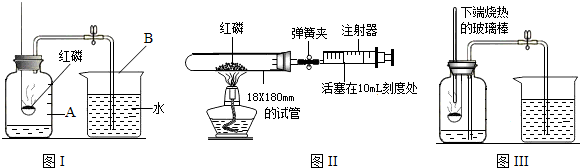
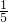 ������Ϊ������һ�����ԭ����________����ֻ��дһ�㣩
������Ϊ������һ�����ԭ����________����ֻ��дһ�㣩