��Ŀ����
ijͬѧΪ�˲ⶨijʯ��ʯ��̼��Ƶ�����������ʯ��ʯ�е����ʲ������ᷴӦ������ȡ25gʯ��ʯ��Ʒ������������ϡ���ᣬͬʱ����5�����������������������������
�Իش��������⣺
��1��������m= g��
��2�������25g��Ʒ��̼��Ƶ���������д��������̣�
��3��25gʯ��ʯ��Ʒ��̼��Ƶ���������Ϊ ��
��4���뻭��ʣ������������ʱ��仯�Ĺ�ϵͼ��
| ʱ��/min | 1 | 2 | 3 | 4 | 5 |
| �������������/g | 2.2 | 4.4 | 6.6 | m | 8.8 |
��1��������m=
��2�������25g��Ʒ��̼��Ƶ���������д��������̣�
��3��25gʯ��ʯ��Ʒ��̼��Ƶ���������Ϊ
��4���뻭��ʣ������������ʱ��仯�Ĺ�ϵͼ��
��������1���������ݱ��в�õ�ʱ�������ӵ��������н��
��2�����ݶ�����̼���������25g��Ʒ��̼��Ƶ��������ɣ�
��3������25g��Ʒ��̼��Ƶ������������25gʯ��ʯ��Ʒ��̼��Ƶ�����������
��4�����ݲμ���Ӧ̼��Ƶ����ʣ�����ʵ�������Ȼ������ʱ���ʣ�������������ɻ���ͼ��
��2�����ݶ�����̼���������25g��Ʒ��̼��Ƶ��������ɣ�
��3������25g��Ʒ��̼��Ƶ������������25gʯ��ʯ��Ʒ��̼��Ƶ�����������
��4�����ݲμ���Ӧ̼��Ƶ����ʣ�����ʵ�������Ȼ������ʱ���ʣ�������������ɻ���ͼ��
����⣺��1����ͼ��֪ÿ�������ɶ�����̼������Ϊ2.2g���ҵ���������ɶ�����̼������Ϊ8.8g�����Ա�����m=8.8g�����8.8��
��2����μӷ�Ӧ��̼��Ƶ�����Ϊx��
CaCO3+2HCl�TCaCl2+H2O+CO2��
100 44
x 8.8g
=
x=20g
��25g��Ʒ��̼��Ƶ�����Ϊ20g��
��3��25gʯ��ʯ��Ʒ��̼��Ƶ���������=
��100%=80%�����80%��
��4��ʣ����������=25g-20g=5g������ʣ������������ʱ��仯�Ĺ�ϵͼ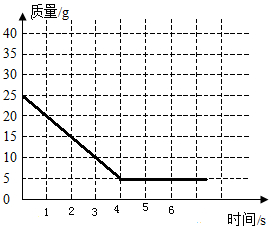 �����
����� ��
��
��2����μӷ�Ӧ��̼��Ƶ�����Ϊx��
CaCO3+2HCl�TCaCl2+H2O+CO2��
100 44
x 8.8g
| 100 |
| x |
| 44 |
| 8.8g |
x=20g
��25g��Ʒ��̼��Ƶ�����Ϊ20g��
��3��25gʯ��ʯ��Ʒ��̼��Ƶ���������=
| 20g |
| 25g |
��4��ʣ����������=25g-20g=5g������ʣ������������ʱ��仯�Ĺ�ϵͼ
 �����
����� ��
�����������ݷ�Ӧ�Ļ�ѧ����ʽ���Ա�ʾ��Ӧ�и����ʵ������ȣ��ɷ�Ӧ��ij�����ʵ������ɼ������Ӧ���������ʵ�������

��ϰ��ϵ�д�
 ��������ϵ�д�
��������ϵ�д� ���ɶ���ܲ��¿�ֱͨ�߿�ϵ�д�
���ɶ���ܲ��¿�ֱͨ�߿�ϵ�д�
�����Ŀ
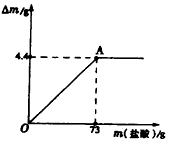
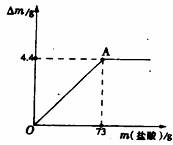
 ��2008?���ݣ���ľ����ũ�峣�õ�һ�ּطʣ�����ֻ������Ч�ɷ�K2CO3������ˮ��ʵ����ѧ��ѧ��ȤС���ͬѧΪ�˲ⶨij��ľ����K2CO3�ĺ�������ȡ�ò�ľ��100g��������ˮ�����ܽ⣮�����ˡ�ϴ�ӣ�ϴҺ������Һ�У�����Һ������80.4gʱֹͣ���Ȳ��������£�����������μ���ϡ���ᣬͬʱ�����ɵ�CO2��������ʯ����[Ca��OH��2]���գ��������������m��ʯ���������ӵ�������m�Ĺ�ϵ��ͼ��ʾ��
��2008?���ݣ���ľ����ũ�峣�õ�һ�ּطʣ�����ֻ������Ч�ɷ�K2CO3������ˮ��ʵ����ѧ��ѧ��ȤС���ͬѧΪ�˲ⶨij��ľ����K2CO3�ĺ�������ȡ�ò�ľ��100g��������ˮ�����ܽ⣮�����ˡ�ϴ�ӣ�ϴҺ������Һ�У�����Һ������80.4gʱֹͣ���Ȳ��������£�����������μ���ϡ���ᣬͬʱ�����ɵ�CO2��������ʯ����[Ca��OH��2]���գ��������������m��ʯ���������ӵ�������m�Ĺ�ϵ��ͼ��ʾ��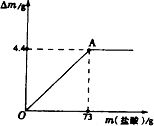
 ��ľ����ũ�峣�õ�һ�ּطʣ�����ֻ������Ч�ɷ�K2CO3������ˮ��ʵ����ѧ��ѧ��ȤС���ͬѧΪ�˲ⶨij��ľ����K2CO3�ĺ�������ȡ�ò�ľ��100g��������ˮ�����ܽ⣮�����ˡ�ϴ�ӣ�ϴҺ������Һ�У�����Һ������80.4gʱֹͣ���Ȳ��������£�����������μ���ϡ���ᣬͬʱ�����ɵ�CO2��������ʯ����[Ca��OH��2]���գ��������������m��ʯ���������ӵ�������m�Ĺ�ϵ��ͼ��ʾ��
��ľ����ũ�峣�õ�һ�ּطʣ�����ֻ������Ч�ɷ�K2CO3������ˮ��ʵ����ѧ��ѧ��ȤС���ͬѧΪ�˲ⶨij��ľ����K2CO3�ĺ�������ȡ�ò�ľ��100g��������ˮ�����ܽ⣮�����ˡ�ϴ�ӣ�ϴҺ������Һ�У�����Һ������80.4gʱֹͣ���Ȳ��������£�����������μ���ϡ���ᣬͬʱ�����ɵ�CO2��������ʯ����[Ca��OH��2]���գ��������������m��ʯ���������ӵ�������m�Ĺ�ϵ��ͼ��ʾ��