��Ŀ����
������������ѧ����
A. �����Ҫ����һЩ��������������Ӷ���Сѹǿ B. �˵�ʹ�ù���Ӧ�������ɴ��
C. ��ͥ��·�б��밲װ����˿��������� D. ��ȼ�ŵ�ľ������Һ��ʯ�����Ƿ�й¶
D ��������A�������Ҫ����һЩ��������������Ӷ���Сѹǿ��ѹǿ�ǵ�λ����ϵ�ѹ������A��ȷ��B���˵�ʹ�ù���Ӧ�������ɴ�ţ��������⣬��B��ȷ��C����ͥ��·�б��밲װ����˿��������أ���ֹ©�������õ��¹ʷ�������C��ȷ��D��������ȼ�ŵ�ľ������Һ��ʯ�����Ƿ�й¶��Һ��ʯ������ȼ�գ���D���� �㾦��ѹǿ�ǵ�λ����ϵ�ѹ����������������Ӷ���Сѹǿ����ͥ��·�а�װ����˿�����������ʮ...����һ��Ӧ�ù㷺�Ľ�����ij��ȤС��Խ�����չ����ϵ���о���
I �о����Ļ�ѧ����
��1�����ڳ����»���������Ӧ�������γ�һ�����ܵ�����Ĥ����ѧ����ʽΪ_________ �� ʵ��ǰ����Ҫ�����ı����Ƚ���______________�����������������ȤС��ͬѧ���������ú�� �����ɴ�С��ͬ��СƬ�����ں���ʵ�顣
��2�������ᡢ�����Һ�ķ�Ӧ
ʵ�� | ���� | ���� | ���� |
һ | ����Ƭ����ϡ ������ | ______________���Թܱ��� | ���������ᷢ����Ӧ����Ӧ���� |
�� | ����Ƭ������ ��������Һ�� | �����������ݲ������Թܱ� �� | ����������������Һ��Ӧ����Ӧ ���� |
�� | ������������ ͭ��Һ�� | �������к�ɫ���ʲ������� Һ�����ɫ | ������ԣ���_____ͭ |
ʵ��һ������Ϊ______________________��ʵ����������ϣ������������ơ�ˮ��Ӧ����ƫ�����ƣ�NaAlO2������������Ӧ�Ļ�ѧ����ʽΪ______________________��ʵ�������������Ľ������_____________������ڡ���С�ڡ���ͭ��
��3��С��ͬѧ��ʵ��һ�е�ϡ���ỻ�ɵ�������������Ũ����ͬ��ϡ���ᣬ��ͬʱ���� ������Ƭ������������ݽ��٣���Ӧ������
�Ա���������ʵ��������롣 ����٣�ϡ�����е������ӶԷ�Ӧ�����дٽ����á� ����ڣ�_____��
Ϊ��֤������Ƿ������Ӧ������ϡ�����м���___________������ĸ�����۲�����
A��Na2SO4 B��Na2CO3 C��NaCl
II �ⶨij����Ʒ�н���������������
������1������������������������Һ��Ӧ�������백ˮ��Ӧ��
������2��AlCl3+3NaOH=Al(OH)3��+3NaCl��AlCl3+3NH3��H2O=Al(OH)3��+3NH4Cl��
��4��С��ͬѧ��ȡ4.62gij��������Ʒ����Ʒ�����ʽ�Ϊ��������������ͼһ��ƿ�У���������ϡ��������ȫ��Ӧ������Ӧ���Һ��ֳ���Һ1����Һ2���ȷݣ��������ʵ�鷽������ͼ������ͨ�����������ⶨ��Ʒ�н�����������������
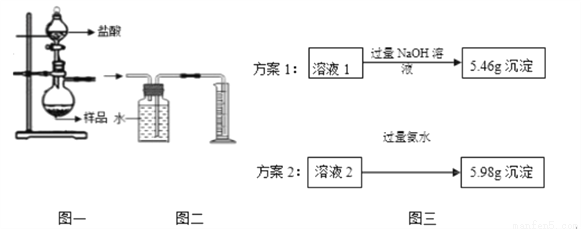
��ѡ����ȷ��һ��ʵ�鷽����������Ʒ�н�������������������д��������̣�.
��С��ͬѧ���ͼһ��ͼ��װ�ò������������ƿ�з�Ӧֹͣ��������Ͳ��ˮ������� ������ֽ���������������ƫ���ܵ�ԭ����?
4Al+3O2=2Al2O3 ��ĥ�������Կɣ� �����������ݲ��� 2Al+2 NaOH + 2H2O =2NaAlO2 + 3H2�� ���� ϡ�����е���������ӶԷ�Ӧ�������������� C �����ÿ������Ԫ�ص����� 2.07g�������������Ʒ����Ԫ�ص����� 4.14g���� ���ÿ�������������� 0.51g�������������Ʒ�������������� 1.02g���� ���ÿ������������ 1.8g��������������Ϊ 77...�����ĸ�ʵ�鷽����Ʋ���������
ѡ�� | ʵ��Ŀ�� | ���� |
A | ������������Һ���������Һ | ȡ�����μ�Ba��NO3��2��Һ���۲����� |
B | ��ȥ�����е����������� | ���������顢�ܽ⡢���ˡ����� |
C | ��ȥCaO�е�CaCO3 | ���¼��ȵ������������� |
D | ̽��Mg��Ag��Cu������ | ������������þ���ֱ����� |


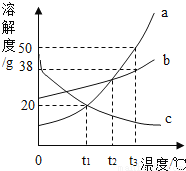
 ϡ��Ũ����
ϡ��Ũ����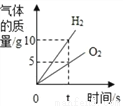 ���ˮһ��ʱ��
���ˮһ��ʱ��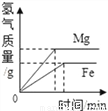 �������� Mg �� Fe ������ͬŨ��ϡ���ᷴӦ
�������� Mg �� Fe ������ͬŨ��ϡ���ᷴӦ Ũ����¶�ÿ�����
Ũ����¶�ÿ�����