��Ŀ����
̩����2013���п���ѧʵ�鿼���У�����������ȡ����������̼����ȡ��������50g 5%��NaCl��Һ������������ǩ,��ѧ����ǩȷ��һ��������п��顣
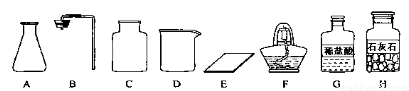
��1����ͬѧ��ǩ������ʵ���ң����ֱ���ʵ��������Ҫ��������������ҩƷ��
����ͼ������A��������??? ��
����ͬѧ�鵽�Ŀ�ǩӦ����????? ������ĸ���ţ���
A����������ȡ?????????? B��������̼����ȡ
����ȡ������ķ�Ӧԭ��Ϊ? ??????????????????? ? ���û�ѧ����ʽ��ʾ������ȡһƿ�����壬Ӧѡ�õ�������?? ??????????? ?? ������ĸ���ţ���
����ͬѧʵ�����Ҫ����ʾ�����£���������������������װ����????????? ������ҩƷ���ռ����塣
�������������巢��װ�û�������ȡ�������壬��д������һ�ַ�Ӧ�Ļ�ѧ����ʽ? ????????? ?? ��
��2����ͬѧ�����������50g 5%��NaCl��Һ��ʵ������У�ͨ����������ȡˮԼ ???? mL������ȡˮʱ���Ӷ��������������Һ����������������? ?????? ������ƫ��������ƫС��������Ӱ���������ܽ�NaClʱ�õ��IJ�������������? ????????????????????????????? ??? ��
��1����? ��ƿ���� B ��? CaCO3+2HCl=CaCl2+H2O+CO2�� ��ABCE ����? ���װ�������ԡ���? 2H2O2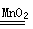 2H2O+ O2����Zn + H2SO4 = ZnSO4 + H2�� ��ֻҪ���������֣�����2��47.5 ��ƫ���裬�ӿ�NaCl���ܽ��ٶ�? ��
2H2O+ O2����Zn + H2SO4 = ZnSO4 + H2�� ��ֻҪ���������֣�����2��47.5 ��ƫ���裬�ӿ�NaCl���ܽ��ٶ�? ��
��������
�����������1������ͼ������A����������ƿ������ҩƷ��������֪���Ŀ�ǩӦ����B������̼����ȡ������ȡ������ķ�Ӧԭ��ΪCaCO3+2HCl=CaCl2+H2O+CO2����Ӧѡ�õ���װ��Һ�����·�Ӧ��װ�ã���������ABCE����ͬѧʵ�����Ҫ����ʾ�����£���������������������װ��������װ�õ�������������ҩƷ���ռ����壻�������������巢��װ�û�������ȡ�������壬����������һ�ַ�Ӧ�Ļ�ѧ����ʽΪZn + H2SO4 = ZnSO4 + H2������2����ͬѧ�����������50g 5%��NaCl��Һ��ʵ������У�ͨ����������ȡˮԼ50g-50g��5%=47.5g��47.5g/1g/Ml=47.5Ml����ȡˮʱ���Ӷ���������ȡ��ˮ�����ƫС�����������Һ�������������������ܽ�NaClʱ�õ��IJ������������ǽ��裬�ӿ�NaCl���ܽ��ٶ�?
���㣺ʵ������ȡ�����˼·�ͷ�������������Һ

 ����ѧ����ϵ�д�
����ѧ����ϵ�д�