��Ŀ����
������ԭ����ͭʵ��õ��Ĺ����ĩΪ����ͭ��ͭ�Ļ�����ѧ��ȤС���ͬѧ�Ը÷�ĩ�е���ͭ�ĺ������вⶨ������ͬѧ��ȡ10�˷�ĩ��Ʒ�����ձ��У�Ȼ����벻ͬ������ͬŨ�ȵ�ϡ���ᣬ��ַ�Ӧ���ձ���ʣ�������г�����ʵ�����ݼ�¼���£�

(1)�����������ݿ�֪����2��ʵ���е�ʣ�������_________(д���������ƻ����)��
(2)��Ʒ�е���ͭ����������Ϊ_________��
(3)�����ϡ������������������Ƕ��٣�(д���������)
�����������⿼����DZ������ݷ����������ݻ�ѧ����ʽ�������������ͭ��ϡ�����Ӧ�����ͭ������ͭ�Ļ������ϡ���ᷴӦʱ����������������������ͭ���������Ƚ�ʵ��1��2��֪��ʵ��1��������ȫ��Ӧ���Ƚ�ʵ��2��3֪��ʵ��2������ͭ��ȫ��Ӧ���ɴ˿�֪����Ʒ��ͭ������Ϊ8 g������Ʒ��ͭ����������Ϊ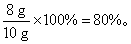
��ʵ��1��20 gϡ��������������Ϊx
CuO + H2SO4====CuSO4+H2O
80 98
10 g-8.4 g x
=1.6 g
80��98=1.6 g��x x=1.96 g
ϡ���� �����ʵ���������
�����ʵ���������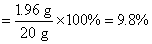
�𰸣�(1)ͭ(��Cu) (2)80%
(3)9.8%(������̼�����)


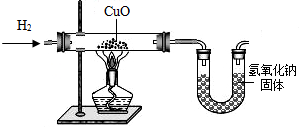
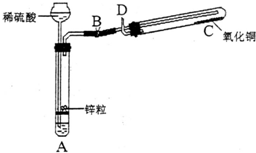 ijѧ������ͼ��ʾװ���Ʊ������������Ƶõ�������ԭ����ͭ��װ���б�Ҫ������̨�����С������豸����ͼ�о�����ȥ��������д���пհף�
ijѧ������ͼ��ʾװ���Ʊ������������Ƶõ�������ԭ����ͭ��װ���б�Ҫ������̨�����С������豸����ͼ�о�����ȥ��������д���пհף�