��Ŀ����
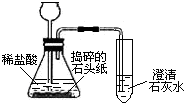 2010��ȫ�������ᡱ������ίԱ���״�ʹ�û�����ʯͷֽ�������ı����ļ�������ʯͷֽ����һ�ֵ�̼���õ���ɫ��Ʒ��������Ҫ�ɷ���һ�ֺ��Ƶ�������ijѧϰС��ԡ�ʯͷֽ����������ͼ��ʾʵ�飬�۲쵽����ʯ��ˮ����ǣ�
2010��ȫ�������ᡱ������ίԱ���״�ʹ�û�����ʯͷֽ�������ı����ļ�������ʯͷֽ����һ�ֵ�̼���õ���ɫ��Ʒ��������Ҫ�ɷ���һ�ֺ��Ƶ�������ijѧϰС��ԡ�ʯͷֽ����������ͼ��ʾʵ�飬�۲쵽����ʯ��ˮ����ǣ�
��ش��������⣺
��1����ʯͷֽ������Ҫ�ɷ���______��
��2��װ��A��B�з�����Ӧ�Ļ�ѧ����ʽΪ��______
��3�������ƹ�ʹ�á�ʯͷֽ�����ɼ���______�������ڻ�������ȫ�������쳣�仯������ЧӦ��
�⣺��1��ʯͷֽ����Ҫ�ɷ���һ�ֺ��Ƶ��������ܹ������ᷴӦ����ʹ����ʯ��ˮ����ǵ����壬��֪ʯͷֽ����Ҫ�ɷ�Ϊ̼��ƣ�
��2�����ݣ�1�����жϿ���ֱ��д����ѧ����ʽ��̼��ƺ����ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼��CaCO3+2HCl�TCaCl2+H2O+CO2�� ������̼���������Ʒ�Ӧ����̼��Ƴ�����ˮ��Ca��OH��2+CO2�TCaCO3��+H2O��
��3��������̼�Ĵ����ŷŻ���������ЧӦ��ֻҪ���������ܹ�����̼�ŷŵģ������ƹ�ʹ�á�ʯͷֽ�����ɼ��� ������̼������ŷţ�
�ʴ�Ϊ����1��̼��ƣ�
��2��CaCO3+2HCl�TCaCl2+H2O+CO2�� Ca��OH��2+CO2�TCaCO3��+H2O
��3��������̼������ŷţ�
��������1�����������֪ʯͷֽ����Ҫ�ɷ���̼��ƣ�
��2��װ��A���������̼��Ƶķ�Ӧ����B���Ƕ�����̼��ʯ��ˮ�ķ�Ӧ������ֱ��д����ѧ����ʽ��
��3���Ӽ��ٶ�����̼���ŷŽǶȷ�����
��������������̼���εĻ�ѧ���ʣ���ס������̼�ļ��鷽��������ȷ��д�����»�ѧ����ʽ��CaCO3+2HCl�TCaCl2+H2O+CO2����Ca��OH��2+CO2�TCaCO3��+H2O��
��2�����ݣ�1�����жϿ���ֱ��д����ѧ����ʽ��̼��ƺ����ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼��CaCO3+2HCl�TCaCl2+H2O+CO2�� ������̼���������Ʒ�Ӧ����̼��Ƴ�����ˮ��Ca��OH��2+CO2�TCaCO3��+H2O��
��3��������̼�Ĵ����ŷŻ���������ЧӦ��ֻҪ���������ܹ�����̼�ŷŵģ������ƹ�ʹ�á�ʯͷֽ�����ɼ��� ������̼������ŷţ�
�ʴ�Ϊ����1��̼��ƣ�
��2��CaCO3+2HCl�TCaCl2+H2O+CO2�� Ca��OH��2+CO2�TCaCO3��+H2O
��3��������̼������ŷţ�
��������1�����������֪ʯͷֽ����Ҫ�ɷ���̼��ƣ�
��2��װ��A���������̼��Ƶķ�Ӧ����B���Ƕ�����̼��ʯ��ˮ�ķ�Ӧ������ֱ��д����ѧ����ʽ��
��3���Ӽ��ٶ�����̼���ŷŽǶȷ�����
��������������̼���εĻ�ѧ���ʣ���ס������̼�ļ��鷽��������ȷ��д�����»�ѧ����ʽ��CaCO3+2HCl�TCaCl2+H2O+CO2����Ca��OH��2+CO2�TCaCO3��+H2O��

��ϰ��ϵ�д�
�����Ŀ
 ��2011?���ױ�����2010��ȫ�������ᡱ������ίԱ���״�ʹ�û�����ʯͷֽ�������ı����ļ�������ʯͷֽ����һ�ֵ�̼���õ���ɫ��Ʒ��������Ҫ�ɷ���һ�������Ը��Σ�ij��ѧ��ȤС���ͬѧ�ԡ�ʯͷֽ���ijɷֽ�������ͼ��ʾ��ʵ��̽�����۲쵽����ʯ��ˮ����ǣ���ش��������⣺
��2011?���ױ�����2010��ȫ�������ᡱ������ίԱ���״�ʹ�û�����ʯͷֽ�������ı����ļ�������ʯͷֽ����һ�ֵ�̼���õ���ɫ��Ʒ��������Ҫ�ɷ���һ�������Ը��Σ�ij��ѧ��ȤС���ͬѧ�ԡ�ʯͷֽ���ijɷֽ�������ͼ��ʾ��ʵ��̽�����۲쵽����ʯ��ˮ����ǣ���ش��������⣺ ��2010?������2010��ȫ�������ᡱ������ίԱ���״�ʹ�û�����ʯͷֽ�������ı����ļ�������ʯͷֽ����һ�ֵ�̼���õ���ɫ��Ʒ��������Ҫ�ɷ���һ�ֺ��Ƶ�������ijѧϰС��ԡ�ʯͷֽ����������ͼ��ʾʵ�飬�۲쵽����ʯ��ˮ����ǣ�
��2010?������2010��ȫ�������ᡱ������ίԱ���״�ʹ�û�����ʯͷֽ�������ı����ļ�������ʯͷֽ����һ�ֵ�̼���õ���ɫ��Ʒ��������Ҫ�ɷ���һ�ֺ��Ƶ�������ijѧϰС��ԡ�ʯͷֽ����������ͼ��ʾʵ�飬�۲쵽����ʯ��ˮ����ǣ�