��Ŀ����
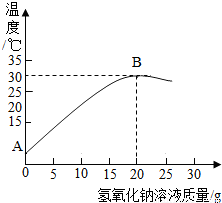
ij��ѧ��ȤС���ͬѧ��ʵ��������������������Ϊ8%������������Һ��������ⶨijϡ���������ʵ�������������ͼ��ʾ�ø�����������Һ��10gijϡ���ᷢ����Ӧ����Һ�¶ȵı仯�������1������150��8%������������Һʱ��Ҫ������______��ȡ______��______��װƿ����ǩ��
��2�����������жϣ��÷�Ӧ������______������Ȼ����ȣ���A�㵽B���������Һ��
pH______��������С��������������������Һ������Ϊ______gʱ����Ӧǡ����ȫ���У�
��3������ϡ���������ʵ�����������
��4������ǡ����ȫ��Ӧʱ������Һ�����ʵ����������Ƕ��٣�
���𰸡���������1��������Һ���Ƶ�һ�㲽��������
��2�����ݷ�Ӧ��������Һ�¶ȵı仯������Ӧ�Ĺ����Լ�ǡ�÷�Ӧ�����ʵ�������
��3�����ݷ�Ӧ�����ĵ��������������������÷���ʽ����Ȼ���������Լ����ɵ��Ȼ��Ƶ�������
��4��������Һ���������Ȼ��Ƶ��������������Ӧ����Һ����������������
����⣺��1��������Һ��һ�㲽���ǣ����㡢��������ȡ����ȡ�������ܽ�������
��2�������������֪��Ӧ��������Һ���¶Ȳ�������ֱ����ȫ��Ӧ�����Ը÷�Ӧ�Ƿ��ȷ�Ӧ����A��B�Ĺ����������������Ƶĵμ�ԭ��Һ�����Բ��ϱ�����pH���ϱ��ֱ������ǡ�÷�Ӧ����Һ��������Һ��pH=7����ͼ���֪����������������Һ������Ϊ20gʱ��Һ���¶���ã��������ƺ�����ǡ�÷�Ӧ��
��3���跴Ӧ��ϡ������������������x�������Ȼ��Ƶ�������y
HCl+NaOH�TNaCl+H2
36.5 40 58.5
10g×x 20g×8% y
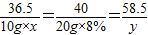
x=14.6% y=2.34g
��4������ǡ����ȫ��Ӧʱ������Һ�����ʵ�����������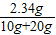 ×100%=7.8%
×100%=7.8%
�ʴ�Ϊ����1�����㣻��ȡ���ܽ⣻��2�����ȣ����20g����3������ϡ���������ʵ���������14.6%����4������ǡ����ȫ��Ӧʱ������Һ�����ʵ�����������7.8%��
�������ɷ�Ӧ���¶ȱ仯���ߣ�������������Һ�ĵμ���Һ�¶������ߣ��¶ȴ���ߵ�ʱ����ʱ��������������ǡ����ȫ��Ӧ��
��2�����ݷ�Ӧ��������Һ�¶ȵı仯������Ӧ�Ĺ����Լ�ǡ�÷�Ӧ�����ʵ�������
��3�����ݷ�Ӧ�����ĵ��������������������÷���ʽ����Ȼ���������Լ����ɵ��Ȼ��Ƶ�������
��4��������Һ���������Ȼ��Ƶ��������������Ӧ����Һ����������������
����⣺��1��������Һ��һ�㲽���ǣ����㡢��������ȡ����ȡ�������ܽ�������
��2�������������֪��Ӧ��������Һ���¶Ȳ�������ֱ����ȫ��Ӧ�����Ը÷�Ӧ�Ƿ��ȷ�Ӧ����A��B�Ĺ����������������Ƶĵμ�ԭ��Һ�����Բ��ϱ�����pH���ϱ��ֱ������ǡ�÷�Ӧ����Һ��������Һ��pH=7����ͼ���֪����������������Һ������Ϊ20gʱ��Һ���¶���ã��������ƺ�����ǡ�÷�Ӧ��
��3���跴Ӧ��ϡ������������������x�������Ȼ��Ƶ�������y
HCl+NaOH�TNaCl+H2
36.5 40 58.5
10g×x 20g×8% y
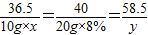
x=14.6% y=2.34g
��4������ǡ����ȫ��Ӧʱ������Һ�����ʵ�����������
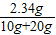 ×100%=7.8%
×100%=7.8%�ʴ�Ϊ����1�����㣻��ȡ���ܽ⣻��2�����ȣ����20g����3������ϡ���������ʵ���������14.6%����4������ǡ����ȫ��Ӧʱ������Һ�����ʵ�����������7.8%��
�������ɷ�Ӧ���¶ȱ仯���ߣ�������������Һ�ĵμ���Һ�¶������ߣ��¶ȴ���ߵ�ʱ����ʱ��������������ǡ����ȫ��Ӧ��

��ϰ��ϵ�д�
�����Ŀ
 12��̼�����ƣ�NaHCO3���׳�С�մ�����ʳƷ��ҽҩ��ҵ��ij��ѧ��ȤС���ͬѧ��̼�����Ƶ����ʽ���̽����
12��̼�����ƣ�NaHCO3���׳�С�մ�����ʳƷ��ҽҩ��ҵ��ij��ѧ��ȤС���ͬѧ��̼�����Ƶ����ʽ���̽����


 ��2012?��̨��ij��ѧ��ȤС���ͬѧ����ͼ��ʾװ�ý���ʵ�飨װ�����������ã����ȹر�ֹˮ�У�������������������Һ������ƿ�У�������յ�������̼���ٴ�ֹˮ�У�
��2012?��̨��ij��ѧ��ȤС���ͬѧ����ͼ��ʾװ�ý���ʵ�飨װ�����������ã����ȹر�ֹˮ�У�������������������Һ������ƿ�У�������յ�������̼���ٴ�ֹˮ�У� ij��ѧ��ȤС���ͬѧ��ʵ����������������Ϊ8%������������Һ��������ⶨijϡ���������ʵ�����������
ij��ѧ��ȤС���ͬѧ��ʵ����������������Ϊ8%������������Һ��������ⶨijϡ���������ʵ�����������