��Ŀ����
��ͼ��ijθҩ��ǩ�ϵIJ������ݣ������ѧ��ѧ��ȤС�����ⶨ��ҩƷ��̼�����ƣ�NaHCO3��������������ȡ10Ƭ��ҩƷ��������ձ��У������ձ��еμ�ϡ������ǡ����ȫ��Ӧ�����ʲ�����ˮ��Ҳ���μӷ�Ӧ����������ϡ����23g����÷�Ӧ���ձ������ʵ�������Ϊ25.8g������㣺��1��10ƬҩƷ������Ϊ______g����Ӧ���������������Ϊ______g��
��2��ҩƷ��̼�����Ƶ�����������
��3����Ӧ��������Һ�����ʵ�������������������������0.1%��

���𰸡���������1������100ƬҩƷ��50g�����ÿƬ��������10Ƭ�������������ձ��������ļ��������Ƕ�����̼����������2�����ݶ�����̼�����������̼�����Ƶ��������ٳ���ҩƷ���������ɣ���3�����ݶ�����̼�����������Ӧ�����ɵ������������ٳ��Է�Ӧ��������Һ���������ɣ�
����⣺��1��100ƬҩƷ��50g�����ÿƬ����Ϊ��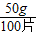 =0.5g������10ƬҩƷ������Ϊ0.5g×10=5g����Ӧ���������������Ϊ5g+23g-25.8g=2.2g��
=0.5g������10ƬҩƷ������Ϊ0.5g×10=5g����Ӧ���������������Ϊ5g+23g-25.8g=2.2g��
��2����Ҫ����2.2g������̼��Ҫ�μӷ�Ӧ��̼����������ΪX�������Ȼ�������ΪY��
NaHCO3+HCl�TNaCl+H2O+CO2��
84 58.5 44
X Y 2.2g
���ݣ�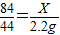 ���X=4.2g������
���X=4.2g������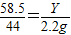 ���Y=2.925g
���Y=2.925g
ҩƷ��̼�����Ƶ���������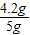 ×100%=84%��
×100%=84%��
��3����Ӧ��������Һ����Ϊ��4.2g+23g-2.2g=25g����Ӧ��������Һ�����ʵ�����������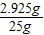 ×100%=11.7%��
×100%=11.7%��
�ʴ�Ϊ����1��5��2.2����2��84%����3��11.7%��
�������������׳����ĵط��Ƿ�Ӧ��������Һ�ļ��㣬���ʲ��ܼ��룬ֻ�ܼ�̼�����ƺ������������ټ�ȥ�����������
����⣺��1��100ƬҩƷ��50g�����ÿƬ����Ϊ��
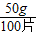 =0.5g������10ƬҩƷ������Ϊ0.5g×10=5g����Ӧ���������������Ϊ5g+23g-25.8g=2.2g��
=0.5g������10ƬҩƷ������Ϊ0.5g×10=5g����Ӧ���������������Ϊ5g+23g-25.8g=2.2g����2����Ҫ����2.2g������̼��Ҫ�μӷ�Ӧ��̼����������ΪX�������Ȼ�������ΪY��
NaHCO3+HCl�TNaCl+H2O+CO2��
84 58.5 44
X Y 2.2g
���ݣ�
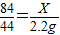 ���X=4.2g������
���X=4.2g������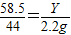 ���Y=2.925g
���Y=2.925gҩƷ��̼�����Ƶ���������
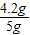 ×100%=84%��
×100%=84%����3����Ӧ��������Һ����Ϊ��4.2g+23g-2.2g=25g����Ӧ��������Һ�����ʵ�����������
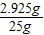 ×100%=11.7%��
×100%=11.7%���ʴ�Ϊ����1��5��2.2����2��84%����3��11.7%��
�������������׳����ĵط��Ƿ�Ӧ��������Һ�ļ��㣬���ʲ��ܼ��룬ֻ�ܼ�̼�����ƺ������������ټ�ȥ�����������

��ϰ��ϵ�д�
�����Ŀ
 ��2012?��������ͼ��ijθҩ��ǩ�ϵIJ������ݣ������ѧ��ѧ��ȤС�����ⶨ��ҩƷ��̼�����ƣ�NaHCO3��������������ȡ10Ƭ��ҩƷ��������ձ��У������ձ��еμ�ϡ������ǡ����ȫ��Ӧ�����ʲ�����ˮ��Ҳ���μӷ�Ӧ����������ϡ����23g����÷�Ӧ���ձ������ʵ�������Ϊ25.8g������㣺
��2012?��������ͼ��ijθҩ��ǩ�ϵIJ������ݣ������ѧ��ѧ��ȤС�����ⶨ��ҩƷ��̼�����ƣ�NaHCO3��������������ȡ10Ƭ��ҩƷ��������ձ��У������ձ��еμ�ϡ������ǡ����ȫ��Ӧ�����ʲ�����ˮ��Ҳ���μӷ�Ӧ����������ϡ����23g����÷�Ӧ���ձ������ʵ�������Ϊ25.8g������㣺 ��ͼ��ijθҩ��ǩ�ϵIJ������ݣ�ij��ѧ��ѧ��ȤС�����ⶨ��ҩƷ��̼�����ƣ�NaHCO3��������������ȡ20Ƭ��ҩƷ��������ձ��У������ձ��еμ�ϡ������ǡ����ȫ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼�����ʲ�����ˮ��Ҳ���μӷ�Ӧ����������ϡ����23g����÷�Ӧ���ձ������ʵ�������Ϊ28.6g��
��ͼ��ijθҩ��ǩ�ϵIJ������ݣ�ij��ѧ��ѧ��ȤС�����ⶨ��ҩƷ��̼�����ƣ�NaHCO3��������������ȡ20Ƭ��ҩƷ��������ձ��У������ձ��еμ�ϡ������ǡ����ȫ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼�����ʲ�����ˮ��Ҳ���μӷ�Ӧ����������ϡ����23g����÷�Ӧ���ձ������ʵ�������Ϊ28.6g��

