��Ŀ����
��10�֣���ѧ�κ�ѧ��ȤС���ͬѧ������ʵ����ʱ��������һƿ����������Һû������Ƥ����������ʦͬ���չ������̽����
[�������1] ������������Һ�Ƿ�������أ�
[ʵ��̽��1]
|
ʵ����� |
ʵ������ |
ʵ����� |
|
ȡ��������Һ���Թ��У�����Һ�еμ�ϡ���ᣬ�������� |
|
����������Һһ�������ˡ� |
[�������2] ������������Һ��ȫ�����ʻ��Dz��ֱ����أ�
[���������]
����1������������Һ���ֱ��ʡ� ����2������������Һȫ�����ʡ�
[��������] �Ȼ�����Һ�����ԡ�
[ʵ��̽��2]
|
ʵ�鲽�� |
ʵ������ |
ʵ����� |
|
��1��ȡ��������Һ���Թ��У�����Һ�еμӹ������Ȼ�����Һ���������� |
�� ���ɡ� |
˵��ԭ��Һ��һ����̼���ơ� |
|
��2��ȡ���裨1���Թ��е������ϲ���Һ���μӷ�̪��Һ�� |
��Һ���ɫ�� |
˵��ԭ��Һ��һ���� �� |
[ʵ�����] ������������Һ ������֡���ȫ���������ʡ�
[��˼������]
������[ʵ��̽��2]�У�С�������������������Һ�����Ȼ�����Һ������Ϊ�÷��� ������С������С�����������
[������Ӧ��] ��1�� ����������Һ���ױ��ʣ������ܷⱣ�档ʵ���ұ����ܷⱣ���ҩƷ���кܶ࣬������һ���� ��
��2��ȡ�������ֱ��ʵ�����������Һ100g�������м�������������������Һ����ȫ��Ӧ��õ�1.97���ɫ����������������������Һ��̼���Ƶ�������������д��������̣�
[ʵ��̽��1]��ÿ��1�֣�
|
ʵ����� |
ʵ������ |
ʵ����� |
|
ȡ��������Һ���Թ��У�����Һ�еμ�ϡ���ᣬ�������� |
������ |
����������Һһ�������ˡ� |
[ʵ��̽��2]
|
ʵ�鲽�� |
ʵ������ |
ʵ����� |
|
��1��ȡ��������Һ���Թ��У�����Һ�еμӹ������Ȼ�����Һ���������� |
�� ��ɫ���� ���ɡ� |
˵��ԭ��Һ��һ����̼���ơ� |
|
��2��ȡ���裨1���Թ��е������ϲ���Һ���μӷ�̪��Һ�� |
��Һ���ɫ�� |
˵��ԭ��Һ��һ���� �������� �� |
[ʵ�����] ������������Һ ���� ������֡���ȫ���������ʡ�
�÷��������� ������С������С���������������������̼���Ʒ�Ӧ�������������� ���𰸺������ɣ�
[������Ӧ��] ��1�� ����������Һ���ױ��ʣ������ܷⱣ�档ʵ���ұ����ܷⱣ���ҩƷ���кܶ࣬������һ���� Ũ���ᣨ�𰸺������ɣ� ��
��2��1.06%
��������
�����������ʵ��̽��һ�У���Ʒ�Ƿ���ʣ�����̽����������̼���ƣ���������������ð��˵������һ������̼���ƣ�˵���Ѿ������ˣ�̽������Ҫ��̽����Ʒ����ȫ���ʾ���̽����Ʒ��ȫ��̼���ƻ���̼���ƺ��������ƵĻ�����������Ȼ������Ȼ�����������ȫ��Ϊ���ԣ�����������Ϊ���ԣ�˵������������ʣ�࣬ԭ��ƷΪ���ֱ��ʣ����Ϊ��������Ʒȫ��Ϊ̼���ƣ���Ʒ��ȫ���ʡ����������������ƴ����Ȼ�������Ϊ�������ƺ�̼���Ʒ�Ӧ����̼��ƺ��������ƣ���ͨ���ⶨ��Ӧ����ҺpHֵ���ⶨ��Ʒ�Ƿ���ȫ���ʡ�������Ӧ���У�ʵ�����ܷⱣ���ҩƷ�ܶ࣬����Ũ���ᣬŨ����ȡ�
�裺��Ʒ��̼���Ƶ�������X
Na2CO3 + Ba(OH)2= BaCO3 + 2NaOH
106 197
X 1.97g
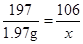

��̼���Ƶ���������Ϊ��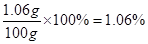
��������������Һ��̼���Ƶ���������Ϊ1.06%
���㣺�������Ƶ�����
�����������Ŀ��Ϊ�ۺϣ��й���ʵ��̽���Ŀ���Ҳ�й��ڼ�����Ŀ��죬�����Ϊ���棬ע����ϸ���⣬ע����Ŀ��С��ǰ��֮�����ϵ��

