��Ŀ����
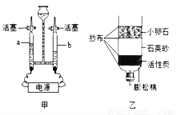
ij��ѧ��ȤС��Ϊ�˲ⶨ���ص�ʯ��ʯ����Ҫ�ɷ���CaCO3��������������������������ʵ�飺ȡ12����Ʒ�����ձ��У�����������120��һ������������ϡ���ᣨ���ʲ�����ˮҲ�����ᷴӦ�������������������ʣ������������ϵ��ͼ��ʾ�������ͼ�е����ݽ��м��㣺

��1��ʯ��ʯ��Ʒ��CaCO3������Ϊ __________�ˣ�
��2�������������������������__________��������ݻ�ѧ����ʽд�������ļ��㲽�裩
10 7.3% ����������1��������ٵ������Dzμӷ�Ӧ��̼��Ƶ�����������ʯ��ʯ��Ʒ��CaCO3������Ϊ��12g-2g=10g����2����ͼ����ǡ�÷�Ӧʱ����ϡ�����������100g����100g�������������ʵ�������x�� CaCO3 + 2HCl == CaCl2 + H2O + CO2�� 100 73 10g x =�����x=7.3g ���������������������Ϊ...
��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
��ȥ���������е�����(������������)�����в�����������ȷ����
ѡ�� | ���� | �������� | |
A | CO(CO2) | ͨ��װ��������ʯ��(NaOH �� CaO)�ĸ���� | |
B | NaNO3(Na2SO4) | ���������� Ba(NO3)2 ��Һ����ַ�Ӧ����� | |
C | CuSO4 ��Һ(����) | ������� CuO ��ĩ����ַ�Ӧ����� | |
D | KCl ��Һ(CaCl2) | ͨ������� CO2 ���壬���� |
A. A B. B C. C D. D
D ��������A����ȥCO��CO2����ͨ��װ��������ʯ�ң�NaOH��CaO���ĸ���ܣ�������̼�ܱ���ʯ�����գ���ȷ��B����ȥNaNO3��Na2SO4��������������Ba��NO3��2��Һ��������ӦΪBa(NO3)2+Na2SO4=BaSO4��+2NaNO3����ȷ��C����ȥCuSO4��Һ�����ᣩ���������CuO�ۣ�������ӦΪCuO+H2SO4=CuSO4+H2O����ַ�Ӧ����ˣ���ȷ��D��CO2����...