��Ŀ����
��λͬѧ�ֱ�����ͬ����������ϡ���ᣬ�ⶨijʯ��ʯ��Ʒ��̼��Ƶ���������(ʯ��ʯ�е����ʼȲ����ᷴӦ��Ҳ������ˮ)��
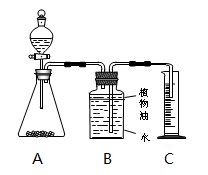
��1��С���ķ��������������̱�ʾ����õ�̼��Ƶ���������Ϊ ���ڹ��˲����У���Ҫ�õ��IJ��������� (ѡ����ţ���
A���ƾ��ơ�©���������� B�� ©�������������ձ� C�� �ƾ��ơ��Թܡ�������
��2��С��ȡ10����Ʒ������ͼ��ʵ��װ�ý���ʵ�飬��Ӧ�������Bװ������������4.6�ˣ������������ݣ����̼��Ƶ�������������������ʵ���ز����������������Ŀ���ԭ���� �ߡ�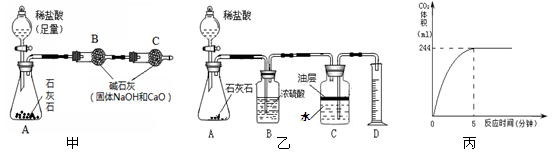
��3��С������ͼ�ҵ�ʵ��װ�ý���ʵ�飬��20��ϡ������뵽1.25����Ʒ�У����ɵ�CO2����뷴Ӧʱ��Ĺ�ϵ��ͼ������֪��ʵ��������CO2���ܶ�Ϊ1.8��/�������ʯ��ʯ��Ʒ��CaCO3������������
��4����ʵ�����õ�ϡ������������������
��1��79%��1�֣��� B��2�֣���(2��B�������˶�����̼������ˮ������HCl���� (1�֣�
(3��80%��(4��7.3%��2�֣�
������������� ��1��ʯ��ʯ�е���Ҫ�ɷ�Ϊ̼��ƣ��������ᷴӦ���ܽ⣻������֪��ʯ��ʯ������Ϊ2.1g����̼�������Ϊ10g-2.1g=7.9g������������Ϊ ��100%=79%�����ݹ��˲�����ԭ����װ�ÿ�֪���ڹ��˲����У���Ҫ�õ��IJ���������B�� ©�������������ձ�����2����Ϊ��������Һ�Ҿ��лӷ��ԣ���������ȡ�Ķ�����̼�к���ˮ�ֺͻӷ������Ȼ������壬��ʯ���е��������ƺ���ʯ�Ҷ�����ˮ�������������Ʒ�Ӧ��ʹBװ���������ӣ���3����֪�����ɵĶ�����̼�����������û�ѧ����ʽ�������δ֪��̼��Ƶ����������岽�����£�
��100%=79%�����ݹ��˲�����ԭ����װ�ÿ�֪���ڹ��˲����У���Ҫ�õ��IJ���������B�� ©�������������ձ�����2����Ϊ��������Һ�Ҿ��лӷ��ԣ���������ȡ�Ķ�����̼�к���ˮ�ֺͻӷ������Ȼ������壬��ʯ���е��������ƺ���ʯ�Ҷ�����ˮ�������������Ʒ�Ӧ��ʹBװ���������ӣ���3����֪�����ɵĶ�����̼�����������û�ѧ����ʽ�������δ֪��̼��Ƶ����������岽�����£�
����CO2��m=��V=1.8g/L��0.244L��0.44g (1�֣�
��μӷ�Ӧ��̼�������ΪX����
CaCO3+2HCl=CaCl2+CO2��+H2O
100 44
X 0.44g
100��44=X��0.44g
X=1g
CaCO3%=1g/1.25g��100%=80%��1�֣�
(4��Ӧ�ҳ�������ȫ��Ӧʱ���������������Ȼ����������������ã���֪��50g���ᣬ�Է�Ӧ��̼�������Ϊ10g-5g=5g�����û�ѧ����ʽ��������Ȼ����������
��μӷ�Ӧ��ϡ�������������ΪY����
CaCO3+2HCl=CaCl2+CO2��+H2O
100 73
5g 50gY
100:73==5g:50gY
Y =7.3%��2�֣�
���㣺 ��ѧ����ʽ�ļ���

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д���13�֣�ij��ѧ����С����ʵ��������ʯ��ˮʱ�������ձ��ײ��й���δ�ܽ⣬����ʯ����Ʒ����Ҫ�ɷֽ�����̽��������������۲����ʵ�鱨�档
��������⡿��Ը���ʯ����Ʒ�ijɷ֣�ͬѧ��չ�������ۣ�С����Ϊ�ܽ��ʣ�����Ϊ���ʺ�����������ʣ����ʵĻ�ѧ����ʽΪ ��С��ͬѧ��ͬ�������жϣ�����ΪС��ͬѧ�������� ��
�����в��롿����һ����ʯ����Ʒ�ɷ���Ca(OH)2��
���������ʯ����Ʒ�ɷ���CaCO3��
������������Ϊ��ʯ����Ʒ�ɷֻ������� ��
��ʵ������ۡ�����ͨ��ʵ����֤��IJ��룺
| ʵ�鲽�� | ʵ������ | ʵ����� |
| ��ȡС�ձ��е����ʽ��� ����������ƣ����õ������Һ�塣 | | ��������ȷ |
| ���������Һ���м������� ��Һ�� | ��Һ��� | |
| ��������ù����м�������ϡ���ᡣ | |
��1��С��ͬѧ��˼����ʯ�ұ��ʵ�ԭ���ڳ��л�ѧ������ѧ���������л��� ����һ������Ҳ�������������ʣ���ʶ�����������ʶ�Ӧ ���档
��2��С����ⶨ��Ʒ��Ca(OH)2��������������ȡ����0.5g��Ʒ��������ˮʹ֮����ܽ⣬������˺���ʵ������pH��ֽ�����ƺõ�10%������Һ����ʵ�飬ʵ�����ݼ�¼���±���
| ����������Һ������/g | 0 | 2.5 | 3.65 | 6 |
| ��Ʒ��pH | 12 | 11 | 7 | 2 |
������Ʒ��Ca(OH)2������������