��Ŀ����
��ѧ�����������ߣ������ǵ������������е���ϵ����1�����е�������������ʯ�ҡ���ʯ�ҡ�̼�����Ƶ����ʣ�����ѡ����ʵ����ʰ�Ҫ����գ�
�ٺ��ӽ���ʱ���������������� ����������ҩƬ����Ҫ�ɷ֣�̼��ơ����ۣ�
�ڳ�����ʳƷ��������� �������ԭ���� ���û�ѧ����ʽ��ʾ����
�ۿ���������θ�������� ��
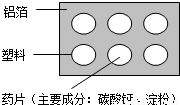
��2����ͼΪij����ҩ��ʵ��ͼ���ش��������⣮
��ͼ�б�ʾ�������У������л��ϳɲ��ϵ��� ��
�ڵ������� ������ĸ��ţ���
A�������� B������ C����֬ D��ά����
��ͼ�����ϰ�װ�������������ʵ�飮���ŵ��� ��
���𰸡���������1�����ʵ����ʾ������ʵ���;�����ݳ������ʵ���������;���з�����ɣ�
��2�����ݲ��ϵķ����������������Ӫ���ط�����
����⣺��1���ٵ�����ѧ���ʲ����ã����ӽ���ʱ�����ڱ��������ʴ�Ϊ��������
����ʯ������ˮ��Ӧ�����������ƣ�������ʳƷ���������Ӧ�Ļ�ѧ����ʽ��CaO+H2O=Ca��OH��2��
�ʴ�Ϊ����ʯ�ң�CaO+H2O=Ca��OH��2��
��̼�������ܺ�θ��������ᷴӦ������������θ����࣬�ʴ�Ϊ��̼�����ƣ�
��2���ٸ���ͼʾ���ѿ����������ǽ������ϣ��������л��߷��Ӻϳɲ��ϣ��ʴ�Ϊ�����ϣ�
�ڵ��������࣬�ʴ�Ϊ��B��
�����ϰ�װ�������������ʵ�飬���ŵ��ǽ�ԼҩƷ���������ԣ����ڶԱȵȣ��ʴ�Ϊ����ԼҩƷ���������ԣ����ڶԱȵȣ�
�����������ѶȲ��Ǻܴ���Ҫ���������ʵ���������;�����ϵķ��ࡢӪ���صȻ�ѧ֪ʶ���Ӷ�����ѧ���������⡢��������������
��2�����ݲ��ϵķ����������������Ӫ���ط�����
����⣺��1���ٵ�����ѧ���ʲ����ã����ӽ���ʱ�����ڱ��������ʴ�Ϊ��������
����ʯ������ˮ��Ӧ�����������ƣ�������ʳƷ���������Ӧ�Ļ�ѧ����ʽ��CaO+H2O=Ca��OH��2��
�ʴ�Ϊ����ʯ�ң�CaO+H2O=Ca��OH��2��
��̼�������ܺ�θ��������ᷴӦ������������θ����࣬�ʴ�Ϊ��̼�����ƣ�
��2���ٸ���ͼʾ���ѿ����������ǽ������ϣ��������л��߷��Ӻϳɲ��ϣ��ʴ�Ϊ�����ϣ�
�ڵ��������࣬�ʴ�Ϊ��B��
�����ϰ�װ�������������ʵ�飬���ŵ��ǽ�ԼҩƷ���������ԣ����ڶԱȵȣ��ʴ�Ϊ����ԼҩƷ���������ԣ����ڶԱȵȣ�
�����������ѶȲ��Ǻܴ���Ҫ���������ʵ���������;�����ϵķ��ࡢӪ���صȻ�ѧ֪ʶ���Ӷ�����ѧ���������⡢��������������

��ϰ��ϵ�д�
 Сѧ���AB��ϵ�д�
Сѧ���AB��ϵ�д� ABC����ȫ�ž�ϵ�д�
ABC����ȫ�ž�ϵ�д�
�����Ŀ