��Ŀ����
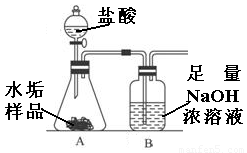
����ʹ�õ�ˮ���ײ�������һ��ˮ��������Ҫ�ɷ���̼��ƺ�������þ��Ϊ��ȷ�ⶨˮ����������þ�ĺ�����ʵ��С��ֱ�ȡ����ͬ����ˮ����Ʒ��7.00g��������ͼ��ʾװ��������������ʵ�飬����ÿ��ʵ����װ��B�������仯��¼���±���
|
|
��һ�� |
�ڶ��� |
������ |
ƽ��ֵ |
|
Bװ�����ӵ�������g�� |
2.17 |
2.22 |
2.21 |
|
��������ʵ�������ݺش�
�ŵ�һ��ʵ�������ݽϵ͵�ԭ���� ��
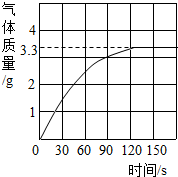
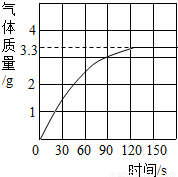
��ƽ��ÿ��ˮ����Ʒ��̼��Ƶ�����Ϊ �ˣ�
��ˮ����Ʒ��������þ��ƽ����������Ϊ ������������ȷ��0.1��
��1��������������٣�û�а�̼��Ʒ�Ӧ�ꣻ��2��5g�� ��3��28.6%
����������1�������������û�н���Ʒ�е�̼���ȫ��Ӧ�����ɵĶ�����̼���٣�B�������ƺͶ�����̼��Ӧ����̼���ƺ�ˮ��������B���ص�����ƫС��
��2������B��Ӧǰ�����ص��������ƽ��ֵ������CaCO3+2HCl=CaCl2+H2O+CO2�����㼴�ɡ�
��3�����ݣ�2���������̼��Ƶ�����������7g��ȥ̼��Ƶ���������������þ�������ٱ���7g���ٷ�֮�ټ��ɽ��
��2������ʵ��ƽ�����ɶ�����̼������Ϊ2.2g��������2.2g��Ҫ̼��Ƶ�����ΪX��
CaCO3+2HCl=CaCl2+H2O+CO2��
44
X 2.2g
���ݣ�100/44=x/2.2g���x=5g��
��3��ˮ����Ʒ��������þ��ƽ����������Ϊ����7.00g-5.00g��/7.00g��100%=28.6%