��Ŀ����
ˮ����Ҫ����Ȼ��Դ����1����ͼ��ʾ��3��ʵ�飬A��ˮ������ �����������ѧ�����仯��B���Թ�1�ڵõ�������Ϊ ��C�о���ˮ�ķ����� ��������
A��ˮ�ķ��� B��ˮ�ĵ�� C��ˮ�ľ���
��2��ʵ���ҵķ�Һ�辭���������ŷţ�Ϊ������������ķ�Һ�����ԣ�����ѡ�õ������� ������ĸ��ţ���
A�������� B����ʯ�� C��������̼ D��̼����
��3������ˮ����������ˮ�Ĺ������������ʯ�ң���ʯ����ˮ��Ӧ�Ļ�ѧ����ʽΪ ����2�֣�
��4�������ƣ�Na2FeO4����һ�����;�ˮ��������������ɱ���������ã��뽫������ȡ�����ƵĻ�ѧ����ʽ����������2Fe��NO3��3+16NaOH+3Cl2=6NaNO3+6NaCl+2Na2FeO4+ ����2�֣�
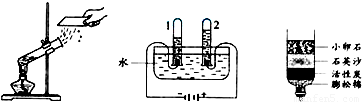
���𰸡���������1�����ݻ�ѧ�仯�������仯�ĸ���Լ����ˮ��ԭ���������ˮ�ľ���ˮ�ķ������н��
��2����������Ļ�ѧ���ʷ������
��3��������ʯ����ˮ��Ӧԭ����д����ʽ��
��4�����ݻ�ѧ��Ӧǰ��ԭ�ӵ������������������
����⣺��1��Aʵ���������������ɣ������������仯��B���Թ�1�ڵõ����������������жϷ����ǣ�һ�������������߶������Թ����ӵ��Ǹ�����Cװ�õĻ���̿���������ԣ��������ʶ�ˮ�й��˺����������ã�
�ʴ�Ϊ��
������������H2�����ˣ�
��2�����������ǰ���������̼���η�Ӧ���������̼����Ӧ��
��ѡC��
��3����ʯ����ˮ��Ӧ�����������ƣ�
�ʴ�Ϊ��CaO+H2O=Ca��OH��2��
��4����ѧ��Ӧǰ��ԭ�ӵ�����������䣬�ɴ˿�֪�÷�Ӧ�ķ���ʽ��2Fe��NO3��3+16NaOH+3Cl2=6NaNO3+6NaCl+2Na2FeO4+8H2O��
�ʴ�Ϊ��8H2O��
���������⿼��֪ʶ�Ƚ�ȫ�棬���ѶȲ����������֪ʶ������ȷ���
��2����������Ļ�ѧ���ʷ������
��3��������ʯ����ˮ��Ӧԭ����д����ʽ��
��4�����ݻ�ѧ��Ӧǰ��ԭ�ӵ������������������
����⣺��1��Aʵ���������������ɣ������������仯��B���Թ�1�ڵõ����������������жϷ����ǣ�һ�������������߶������Թ����ӵ��Ǹ�����Cװ�õĻ���̿���������ԣ��������ʶ�ˮ�й��˺����������ã�
�ʴ�Ϊ��
������������H2�����ˣ�
��2�����������ǰ���������̼���η�Ӧ���������̼����Ӧ��
��ѡC��
��3����ʯ����ˮ��Ӧ�����������ƣ�
�ʴ�Ϊ��CaO+H2O=Ca��OH��2��
��4����ѧ��Ӧǰ��ԭ�ӵ�����������䣬�ɴ˿�֪�÷�Ӧ�ķ���ʽ��2Fe��NO3��3+16NaOH+3Cl2=6NaNO3+6NaCl+2Na2FeO4+8H2O��
�ʴ�Ϊ��8H2O��
���������⿼��֪ʶ�Ƚ�ȫ�棬���ѶȲ����������֪ʶ������ȷ���

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ


