摘要:① CuCuSO4
网址:http://m.1010jiajiao.com/timu_id_920958[举报]
某课外兴趣小组利用废铜制取硫酸铜,设计方案如下
①Cu CuO
CuO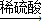 CuSO4
CuSO4
②Cu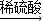 CuSO4
CuSO4
③Cu→CuSO4(Cu+2H2SO4(浓) CuSO4+SO2↑+2H2O)
CuSO4+SO2↑+2H2O)
④Cu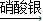 Cu(NO3)2
Cu(NO3)2 Cu(OH)2
Cu(OH)2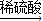 CuSO4
CuSO4
请从理论、操作、环保等方面考虑,你认为其中最合理的是( )
A.①
B.②
C.③
D.④
查看习题详情和答案>>
①Cu
 CuO
CuO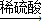 CuSO4
CuSO4②Cu
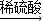 CuSO4
CuSO4③Cu→CuSO4(Cu+2H2SO4(浓)
 CuSO4+SO2↑+2H2O)
CuSO4+SO2↑+2H2O)④Cu
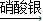 Cu(NO3)2
Cu(NO3)2 Cu(OH)2
Cu(OH)2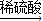 CuSO4
CuSO4请从理论、操作、环保等方面考虑,你认为其中最合理的是( )
A.①
B.②
C.③
D.④
查看习题详情和答案>>
某课外兴趣小组利用废铜制取硫酸铜,设计方案如下
①Cu 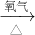 CuO
CuO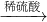 CuSO4
CuSO4
②Cu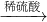 CuSO4
CuSO4
③Cu→CuSO4(Cu+2H2SO4(浓) CuSO4+SO2↑+2H2O)
CuSO4+SO2↑+2H2O)
④Cu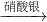 Cu(NO3)2
Cu(NO3)2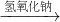 Cu(OH)2
Cu(OH)2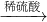 CuSO4
CuSO4
请从理论、操作、环保等方面考虑,你认为其中最合理的是
- A.①
- B.②
- C.③
- D.④
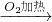 CuO
CuO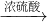 CuSO4
CuSO4 CuSO4+SO2↑+2H2O)
CuSO4+SO2↑+2H2O)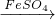 CuSO4
CuSO4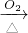 CuO
CuO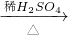 CuSO4
CuSO4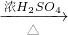 CuSO4;(Cu+2H2SO4(浓)
CuSO4;(Cu+2H2SO4(浓) CuSO4+SO2↑+2H2O)
CuSO4+SO2↑+2H2O)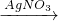 Cu(NO3)2
Cu(NO3)2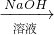 Cu(OH)2
Cu(OH)2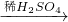 CuSO4
CuSO4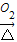 CuO
CuO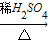 CuSO4
CuSO4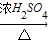 CuSO4;(Cu+2H2SO4(浓)
CuSO4;(Cu+2H2SO4(浓)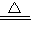 CuSO4+SO2↑+2H2O)
CuSO4+SO2↑+2H2O) Cu(NO3)2
Cu(NO3)2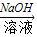 Cu(OH)2
Cu(OH)2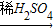 CuSO4
CuSO4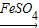 CuSO4
CuSO4