题目内容
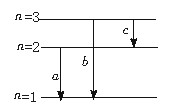
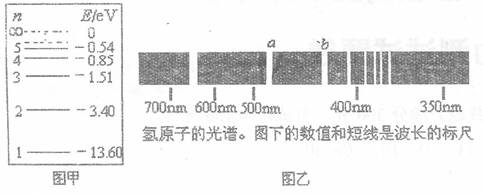
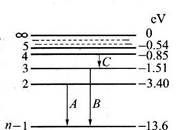
用一束单色光照射处于基态的一群氢原子,这些氢原子吸收光子后处于激发态,并能发射光子,现测得这些氢原子发射的光子频率仅有三种,分别为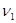 .
.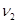 和
和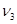 ,且
,且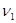 <
<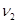 <
<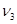 。则入射光子的能量应为( )
。则入射光子的能量应为( )
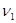 .
.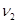 和
和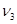 ,且
,且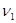 <
<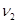 <
<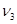 。则入射光子的能量应为( )
。则入射光子的能量应为( )A.h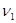 | B.h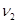 |
C.h(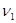 + +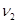 ) ) | D.h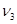 |
CD
本题考查氢原子能级公式和跃迁
根据氢原子能放出三种频率光,判断此时氢原子处在第几能级,然后计算从基态跃迁到该能级需要多少能量,计算能量时根据能级之简能量差和放出光子能量之间的关系计算.同时明确频率、波长、光速之间关系.
能放出三种光,说明此时氢原子处在第3能级,从第三能级跃迁到基态时放出光子能量为:E= h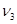 ,或者E= h(
,或者E= h(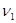 +
+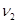 ).所以入射光子能量为E= h
).所以入射光子能量为E= h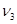 ,或者E= h(
,或者E= h(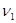 +
+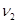 )。
)。
根据氢原子能放出三种频率光,判断此时氢原子处在第几能级,然后计算从基态跃迁到该能级需要多少能量,计算能量时根据能级之简能量差和放出光子能量之间的关系计算.同时明确频率、波长、光速之间关系.
能放出三种光,说明此时氢原子处在第3能级,从第三能级跃迁到基态时放出光子能量为:E= h
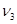 ,或者E= h(
,或者E= h(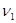 +
+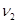 ).所以入射光子能量为E= h
).所以入射光子能量为E= h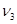 ,或者E= h(
,或者E= h(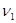 +
+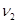 )。
)。
练习册系列答案
 阅读快车系列答案
阅读快车系列答案
相关题目
 ?
?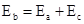 ?
?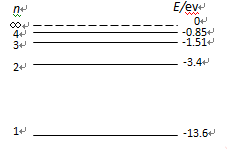
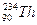 是不稳定的,能自发地发生衰变。
是不稳定的,能自发地发生衰变。 应方程
应方程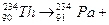 。
。 ,共经过 次
,共经过 次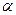 衰变, 次
衰变, 次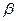 衰变。
衰变。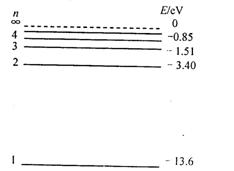
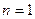 、
、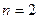 的轨道上运动时,下列相关物理量的关系正确的有 ( )
的轨道上运动时,下列相关物理量的关系正确的有 ( )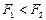
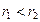
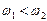
 外电子:( )
外电子:( ) 能增加,电势能减少,动能的增加量小于电势能的减少量
能增加,电势能减少,动能的增加量小于电势能的减少量
 的减少量大于电势能的增加量
的减少量大于电势能的增加量