��Ŀ����
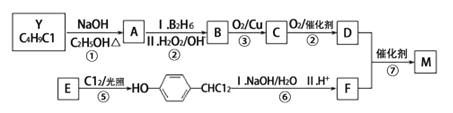
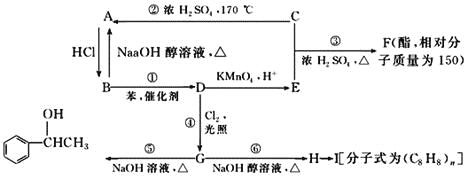
����Ŀ����AΪԭ�Ϻϳ�һ���������ಡҩ����м���M��������ͼ��ʾ��
��֪��
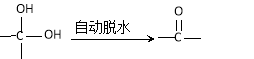
��.ROH + R��X![]() ROR��+ HX ��X��Cl��Br��I��
ROR��+ HX ��X��Cl��Br��I��
��.ȩ��һ�������¿��Է�������ת����
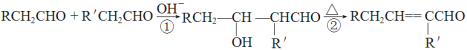
��ش��������� ��
��1����֪��Ӧ��Ϊ�ӳɷ�Ӧ������X�Ľṹ��ʽΪ___________��
��2��������Y�к�����Ԫ�أ�����Y�Ľṹ��ʽΪ___________��
��3��1molG������___________mol Br2��Ӧ��
��4��C����D�Ļ�ѧ��Ӧ����ʽΪ___________________________��
��5��HΪG��ͬ���칹�壬ͬʱ��������������H�Ľṹ��ʽΪ_________��
A������FeCl3������ɫ��Ӧ
B���˴Ź���������ʾ5���
C��1molG��һ���������������3mol NaOH��Ӧ
��6�����������ϳ�·�ߣ��������D�ϳ�E��·�ߡ�
���𰸡���1��HCHO
��2��![]()
��3��2
��4��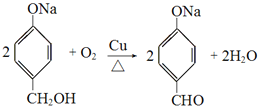
��5��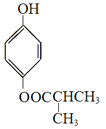
��6��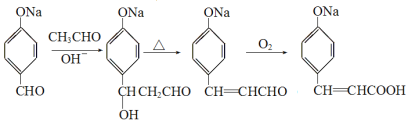
��������
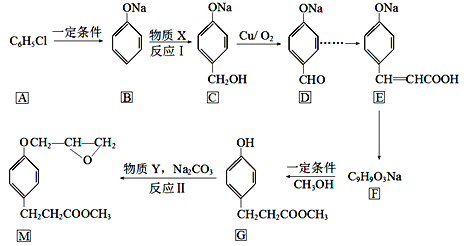
������������������Ϣ��ת����ϵ�ƶϣ�AΪ![]() ��XΪHCHO��YΪ
��XΪHCHO��YΪ![]() ��
��
��1����֪��Ӧ��Ϊ�ӳɷ�Ӧ������X�Ľṹ��ʽΪHCHO��
��2��������Y�к�����Ԫ�أ�����Y�Ľṹ��ʽΪ![]() ��
��
��3�����ǻ���λ����λ�ϵ��ױ���ˮ�е���ԭ��ȡ����1molG������2mol Br2��Ӧ��
��4��C����DΪ���ǻ���ͭ���������ȵ������±���������Ϊȩ������ѧ��Ӧ����ʽΪ ��
��
��5��G�ķ���ʽΪC10H10O3��A������FeCl3������ɫ��Ӧ�����з��ǻ���B���˴Ź���������ʾ5���������5����ԭ�ӣ�C��1molG��һ���������������3mol NaOH��Ӧ��H�Ľṹ��ʽΪ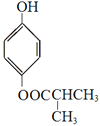 ��
��
��6�����������Ϣ�����D�ϳ�E��·��������