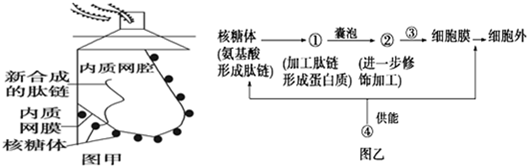
��Ŀ����
6��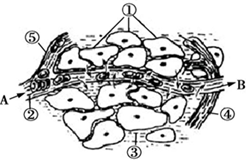 ��ͼΪ����ϸ�����ڻ���֮������ʽ���ʾ��ͼ����ͼ�ش��������⣮
��ͼΪ����ϸ�����ڻ���֮������ʽ���ʾ��ͼ����ͼ�ش��������⣮��1����ͼ��ʾϸ������Χ�����Ĺ�ϵ������ëϸѪ�ܱ�ϸ�������ֱ���ڻ����Ǣڢۣ����ţ���
��2��������ѪҺ������֯ϸ���ķ�ʽ��������ɢ����ϸ����Я��������������Ҫ����6������Ĥ���ܱ���֯ϸ�������ã�
��3��һ������£��ں͢��ڳɷ��ϵ���Ҫ�����Ǣ��к������ʽ϶࣮
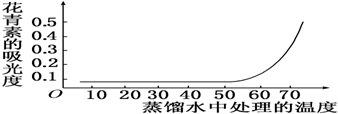
��4���ҹ����ڷ��ֶ���ͷ�����ġ���ͷ�����ޣ����ǵ���ʳ�ǵ����ʺ������µ������̷ۣ���ͷ��������Ҫԭ����ͼ�Т۵�Һ�����࣮
��5��ֱ�Ӳ���ά���ڻ����ȶ����Ĵ�ϵͳ ��ѭ��ϵͳ������ϵͳ������ϵͳ������ϵͳ��
���� ������ͼ��ͼʾΪ����ϸ�����ڻ���֮������ʽ���ʾ��ͼ�����Т�Ϊ��֯ϸ������ΪѪ������Ѫϸ��ֱ������Ļ�������Ϊ��֯Һ������֯ϸ��ֱ������Ļ�������Ϊ�ܰͣ����ܰ�ϸ��������ϸ��ֱ������Ļ�������Ϊ��ϸ����A��ëϸѪ�ܵĶ����ˣ�B��ëϸѪ�ܵľ����ˣ�
��� �⣺��1��ëϸѪ�ܹܱ�ϸ������ľ����ڻ�����Ѫ������֯Һ����ͼ�еĢڢۣ�
��2������������֯ϸ���ķ�ʽ��������ɢ��Ѫ���к�ϸ��Я��������Ҫ������ϸ��Ĥ��ëϸѪ�ܱ�ϸ����2��Ĥ������֯ϸ��Ĥ��������Ĥ��2�㣩�������������ڱ������������������ã���������6��Ĥ��
��3��Ѫ������֯Һ��ȣ�Ѫ���к��н϶����ӵĵ����ʣ���֯Һ�е����ʵĺ������٣�
��4��Ӫ����������ʹѪ��������Ѫ����Ѫ����ѹ���ͣ���֯Һ��ˮ���ͣ��γ���֯ˮ�ף�������ӦҲ���������֯ˮ�ף�
��5��ֱ�Ӳ���ά���ڻ����ȶ����Ĵ�ϵͳ��ѭ��ϵͳ������ϵͳ������ϵͳ������ϵͳ��
�ʴ�Ϊ��
��1���ڢ�
��2��������ɢ��6
��3�����к������ʽ϶�
��4����
��5��ѭ��ϵͳ������ϵͳ������˳��ɵߵ���
���� �����֪ʶ�����ڻ�������ɣ�Ѫ������֯Һ���ܰ�����ɳɷ��ϵIJ�ͬ���ڻ������������ʣ����ʿ�Ĥ����ķ�ʽ�����������Ĺ��̺ͳ�����������ͼ��ȡ��Ϣ�ǽ����ͻ�ƿڣ��������֪ʶ����ۺ�Ӧ���ǽ���Ĺؼ���

| A�� | ������ֻ�а����ϸ���ڴ���ԭ������ | |
| B�� | ��ϸ������ת�ƣ�������ϸ��������ǵ������࣬ϸ��֮�����������ǿ | |
| C�� | PLGF����Ļ�ѧ�����ǵ����ʣ��������ֱ����ص�ϸ�����Ǹ߶������������� | |
| D�� | �ÿ����������ڵ�̥���������ӽ������ϸ������ |
| A�� | �������� | |
| B�� | ���ڵIJ�λ�������ԣ��ͷŵIJ�λ�Ǵ��� | |
| C�� | ������ | |
| D�� | ���ڵIJ�λ�Ǵ��壬�ͷŵIJ�λ�������� |
| A�� | Fe Mn Zn Mg | B�� | Zn Cu Mn Ca | C�� | Zn Cu B Fe | D�� | Mg Mn Cu Mo |
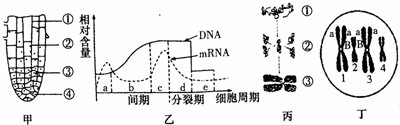 ���м�ͼ��ʾ��и���IJ�ͬ������ͼ��ʾ��и������˿���Ѹ���ϸ������DNA��ϸ������mRNA�����仯����ͼ���ֵ���ϸ�����ѹ����е�һ�����ʱ仯����̬�仯����ͼ��ϸ������ijһʱ��Ⱦɫ�����̬��������ش��������⣺
���м�ͼ��ʾ��и���IJ�ͬ������ͼ��ʾ��и������˿���Ѹ���ϸ������DNA��ϸ������mRNA�����仯����ͼ���ֵ���ϸ�����ѹ����е�һ�����ʱ仯����̬�仯����ͼ��ϸ������ijһʱ��Ⱦɫ�����̬��������ش��������⣺