��Ŀ����
����ʹ�õ�ȼ�ϣ��ִ����ú����Һ��ʯ������ú������Ҫ�ɷ���һ����̼�������Ļ����������ú̿��ˮ(����)��Ӧ�Ƶã����ֳ�ˮú����
(1)��д����ȡˮú������Ҫ��ѧ����ʽ______________________��
(2)Һ��ʯ��������Ҫ�ɷ��DZ��飬����ȼ�յ��Ȼ�ѧ����ʽΪ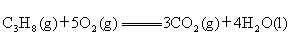 ��
�� ��֪CO����ȼ�յ��Ȼ�ѧ����ʽΪ
��֪CO����ȼ�յ��Ȼ�ѧ����ʽΪ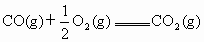 ��
��
 �ԱȽ�ͬ���ʵ�����
�ԱȽ�ͬ���ʵ�����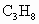 ��COȼ�գ�������������ֵԼΪ______________��
��COȼ�գ�������������ֵԼΪ______________��
(3)��֪����ȼ�յ��Ȼ�ѧ����ʽΪ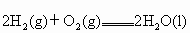 ��
��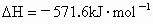 ���ԱȽ�ͬ�����������ͱ���ȼ�գ�������������ֵԼΪ______________��
���ԱȽ�ͬ�����������ͱ���ȼ�գ�������������ֵԼΪ______________��
(4)������δ������Դ����������������֮�⣬�����е��ŵ���_________��
������
|
(1) (2)39 ��5(3)14 ��5(4) ��Դ�ḻ����λ������������������Ⱦ |
��ʾ��
|
�����ϵ�ǰ����ʹ�õ�ȼ�ϣ�������ѧ�����Ȼ�ѧ����ʽ���������㣬������ӱ��֪ʶ�������Ѷ�ϵ����С��ѧ����ϸ�ģ������������⣬Ӧ���ܸ�������𰸣� |

 ��Ӣ���㿨ϵ�д�
��Ӣ���㿨ϵ�д� Ӧ����㲦ϵ�д�
Ӧ����㲦ϵ�д� ״Ԫ����ϵ�д�
״Ԫ����ϵ�д� ͬ������ϵ�д�
ͬ������ϵ�д���8�֣�����ʹ�õ�ȼ�ϣ��ִ����ú����Һ��ʯ������ú������Ҫ�ɷ���CO��H2�Ļ�����壬����ú̿��ˮ������Ӧ�Ƶã����ֳ�ˮú����
��1����д����ȡˮú������Ҫ��ѧ����ʽ____________________________.
��2��Һ��ʯ��������Ҫ�ɷ��DZ��飬����ȼ�յ��Ȼ�ѧ����ʽΪ
C3H8(g) +5O2(g)
== 3CO2(g) + 4H2O(l)����H=�C2220.0 kJ��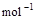 ,
,
��֪CO����ȼ�յ��Ȼ�ѧ����ʽΪ��
CO(g) +1/2O2(g) == CO2(g) ����H=�C282.57kJ��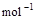 ,
,
�ԱȽ�ͬ������C3H8��COȼ�գ�������������ֵԼΪ_________��1��
��3����֪����ȼ�յ��Ȼ�ѧ����ʽΪ2H2(g)
+ O2(g) == 2H2O(l)����H=�C571.6 kJ��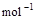 ,�ԱȽ�ͬ������H2��C3H8ȼ�գ�������������ֵԼΪ____________��1��
,�ԱȽ�ͬ������H2��C3H8ȼ�գ�������������ֵԼΪ____________��1��
��4��������δ������Դ������Դ�ḻ֮�⣬�����е��ŵ���____________________

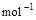 ,�ԱȽ�ͬ������H2��C3H8ȼ�գ�������������ֵԼΪ____________��1��
,�ԱȽ�ͬ������H2��C3H8ȼ�գ�������������ֵԼΪ____________��1�� ,
,