题目内容
已知在A.C8H18(l)+![]() O2(g)
O2(g)![]() 8CO2(g)+9H2O(g) ΔH=-48.40 kJ·mol-1
8CO2(g)+9H2O(g) ΔH=-48.40 kJ·mol-1
B.C8H18(l)+![]() O2(g)
O2(g)![]() 8CO2(g)+9H2O(g) ΔH=-5 518 kJ·mol-1
8CO2(g)+9H2O(g) ΔH=-5 518 kJ·mol-1
C.C8H18(l)+![]() O2(g)
O2(g)![]() 8CO2(g)+9H2O(g) ΔH=+5 518 kJ·mol-1
8CO2(g)+9H2O(g) ΔH=+5 518 kJ·mol-1
D![]() 16CO2(g)+18H2O(g) ΔH=-11 036 kJ·mol-1
16CO2(g)+18H2O(g) ΔH=-11 036 kJ·mol-1
BD
解析:1 mol C8H18燃烧放出热量为48.40 kJ/g×

练习册系列答案
 阅读快车系列答案
阅读快车系列答案
相关题目
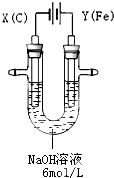 高铁酸钾(K2FeO4)具有高效的消毒作用,为一种新型非氯高效消毒剂.电解法制备高铁酸钾操作简便,成功率高,易于实验室制备.其原理如图所示.
高铁酸钾(K2FeO4)具有高效的消毒作用,为一种新型非氯高效消毒剂.电解法制备高铁酸钾操作简便,成功率高,易于实验室制备.其原理如图所示.